Invasive brain-computer interfaces (BCIs) have long faced a critical challenge: how to achieve stable recording over months or even longer durations while ensuring signal quality. Although cortical surface electrodes and intracortical microelectrodes can achieve high signal-to-noise ratios, chronic implantation is often accompanied by persistent immune responses, tissue remodeling, and mechanical micromotion, leading to gradual signal degradation. While endovascular BCIs developed in recent years have reduced surgical trauma to some extent, vascular responses and long-term safety remain challenges.
Addressing this issue, the research teams led by Professor Xiaorong Gao and Professor Ran Liu from the School of Biomedical Engineering at Tsinghua University, Professor Bai Lu from the School of Pharmaceutical Sciences, and Professor Xiaogang Chen from the Chinese Academy of Medical Sciences, published a research paper online in the journal National Science Review on February 5, 2026, titled "Lateral Ventricular Brain-Computer Interface System with Lantern-Inspired Electrode for Stable Performance and Memory-Guided Decoding."
This study proposes a novel BCI approach based on the lateral ventricular pathway and designs a lantern-inspired flexible electrode that can be deployed within the ventricle. This electrode achieves long-term stable neural signal recording and demonstrates superior decoding capability compared to traditional cortical electrodes in memory-guided decision-making tasks.
Click here for more paper information
Instead of continuing to optimize the cortical interface, the research pivoted to an anatomical location, turning attention to the lateral ventricle, a cerebrospinal fluid-filled cavity structure. The interior of the lateral ventricle features low cell density and a relatively mild mechanical environment, while being spatially adjacent to the cortex and deep structures like the hippocampus and thalamus. Concurrently, external ventricular drainage pathways are relatively mature in clinical practice, providing a practical basis for device delivery.
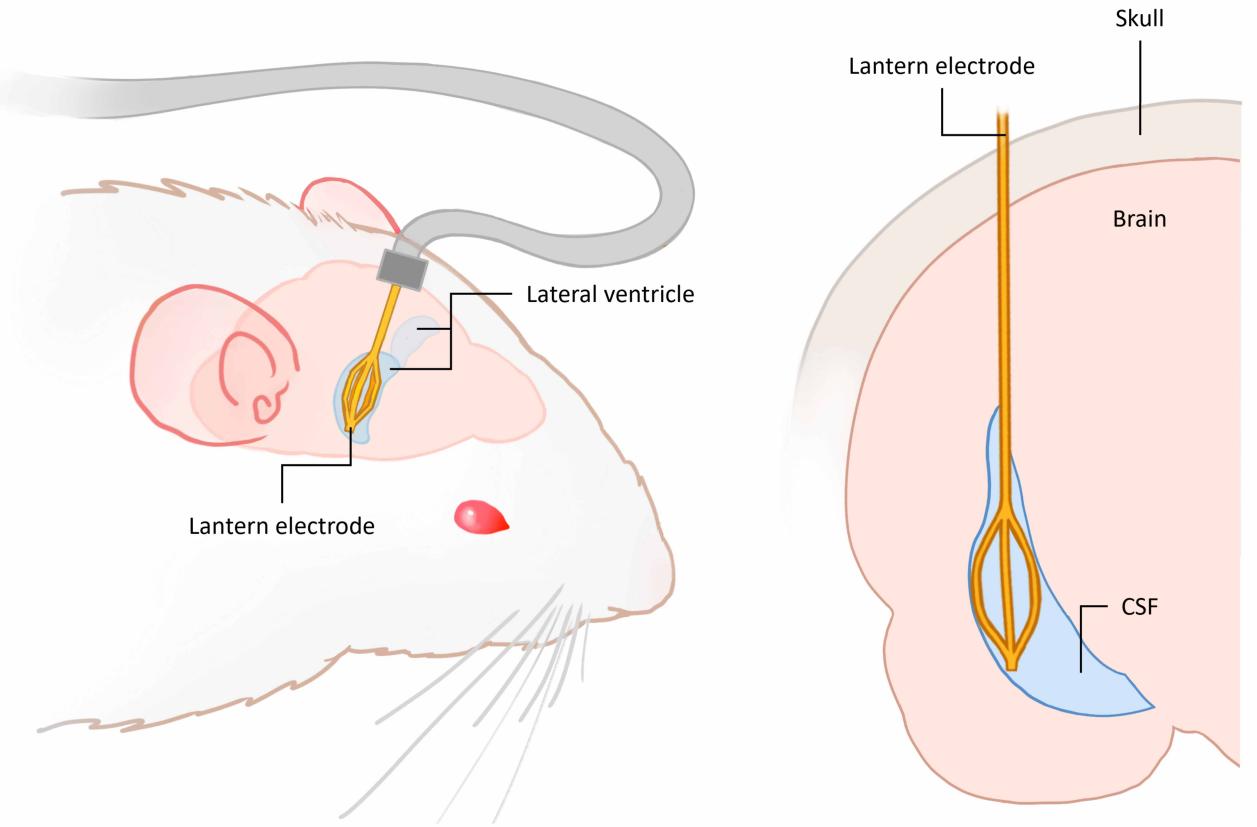
Schematic diagram of lateral ventricular brain-computer interface implantation
Centered on this concept, the research team designed a lantern-inspired expandable flexible electrode. During implantation, the electrode remains in a collapsed state, and after being delivered through a narrow channel to the target location, it radially expands to form a gentle conformal contact with the ependymal wall. The structural design draws inspiration from the force-bearing mechanism of a lantern, using bow-shaped support units to disperse contact stress and reduce mechanical irritation to the ependyma. Finite element analysis results indicate that, compared to rigid structures, this design significantly reduces the deformation level of the ependymal wall and maintains material stress within a safe range during deployment.
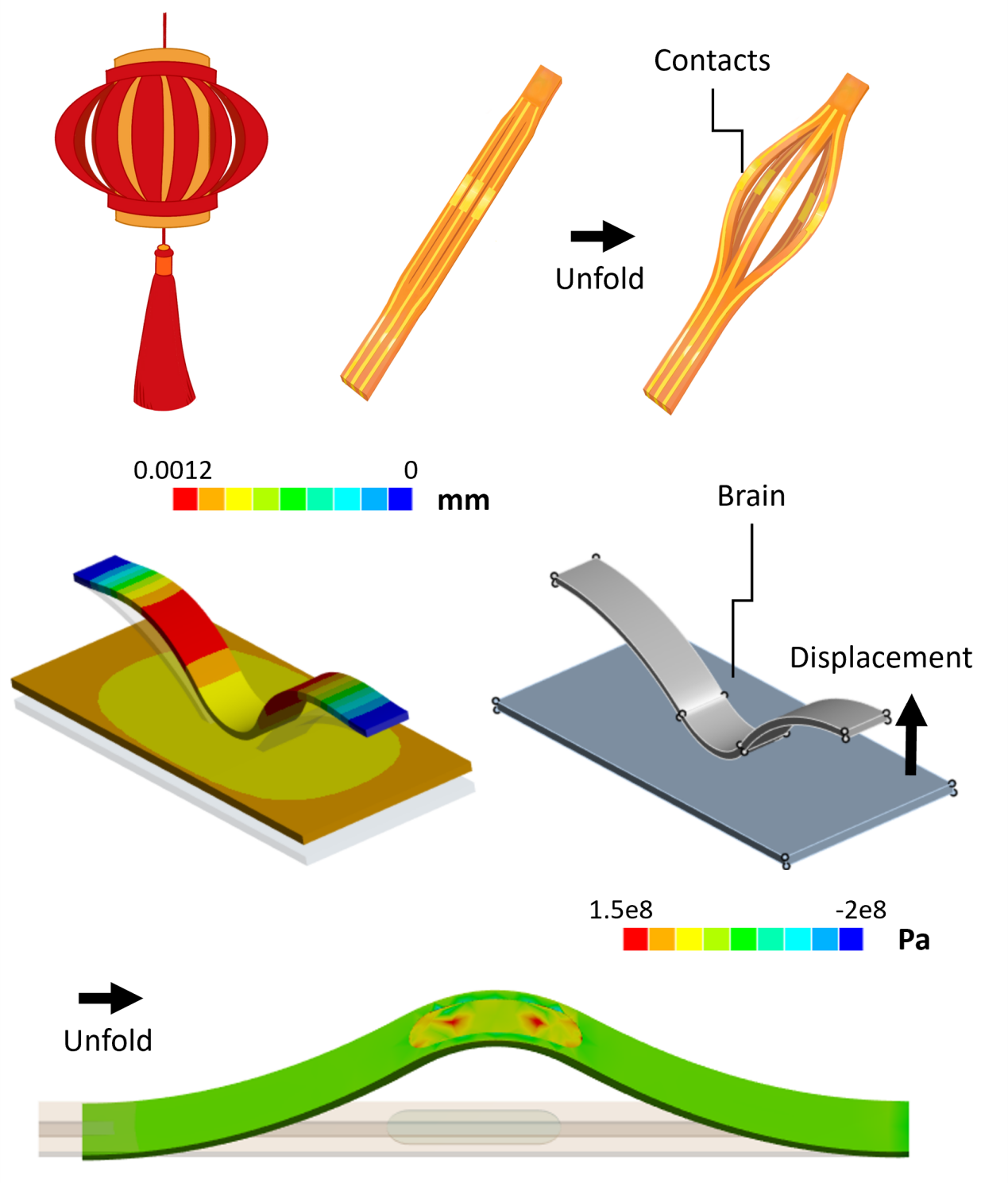
Electrode design and mechanical analysis
In long-term animal experiments, the research team compared the ventricular electrode with electrocorticography (ECoG) electrodes made of the same material. Resting-state spectral analysis showed comparable frequency coverage between the two electrode types, indicating that the ventricular location did not significantly limit the ability to acquire high-frequency information. In visual and auditory steady-state evoked potential experiments, cortical electrodes exhibited higher signal-to-noise ratios in the early post-operative period but showed a gradual decline over time. In contrast, the ventricular electrode maintained stable performance over several months of observation, with signal quality surpassing the control group in later stages.
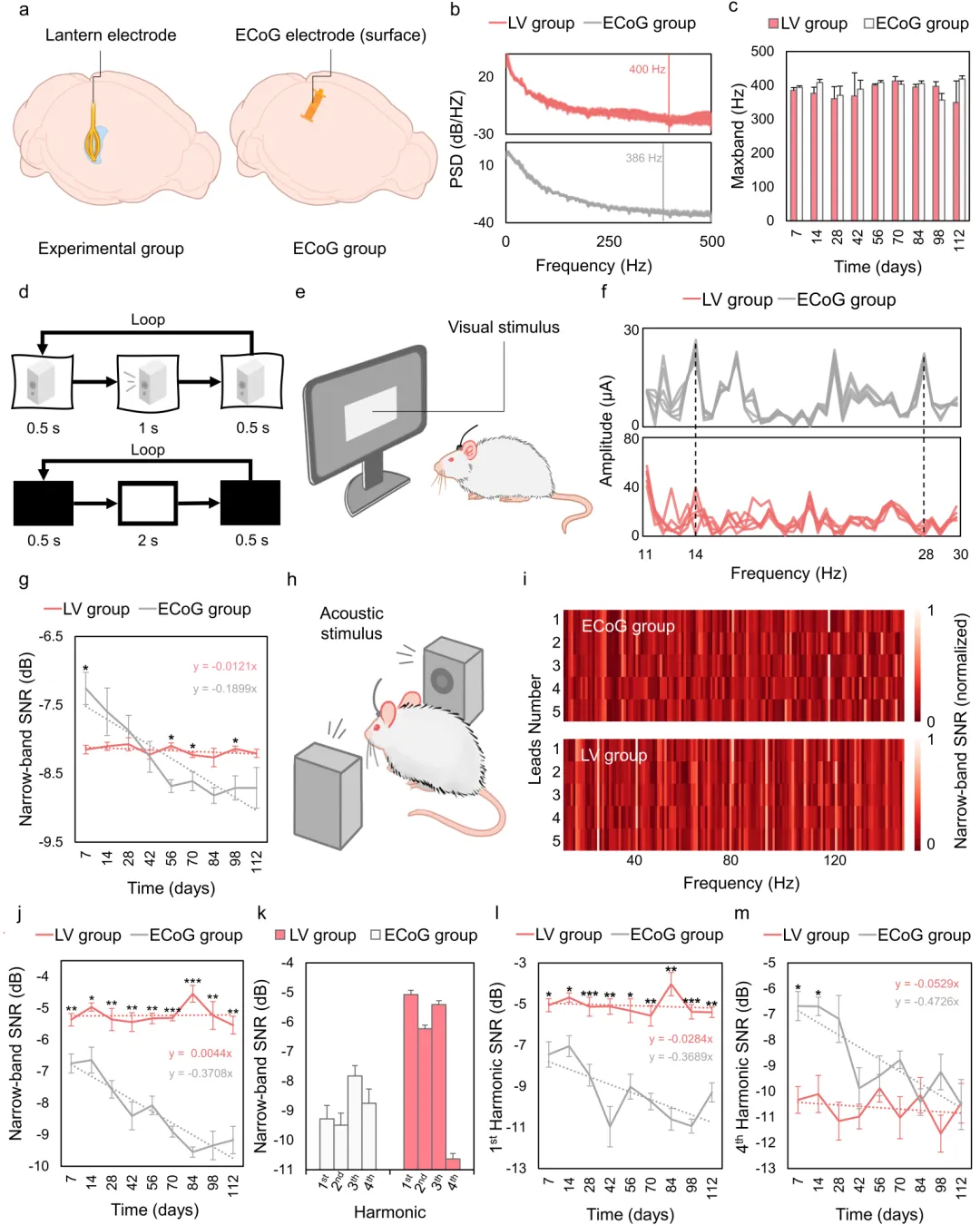
Results of long-term electrical signal recording experiments
Immunohistochemistry results further revealed persistent microglial activation surrounding the cortical electrodes, whereas the ventricular electrode only elicited a transient response in the early post-operative period, subsequently returning to near-control levels. These results suggest that placing the electrode within the cerebrospinal fluid environment and reducing direct mechanical coupling with brain parenchyma helps mitigate chronic inflammatory responses, thereby supporting long-term stable recording.
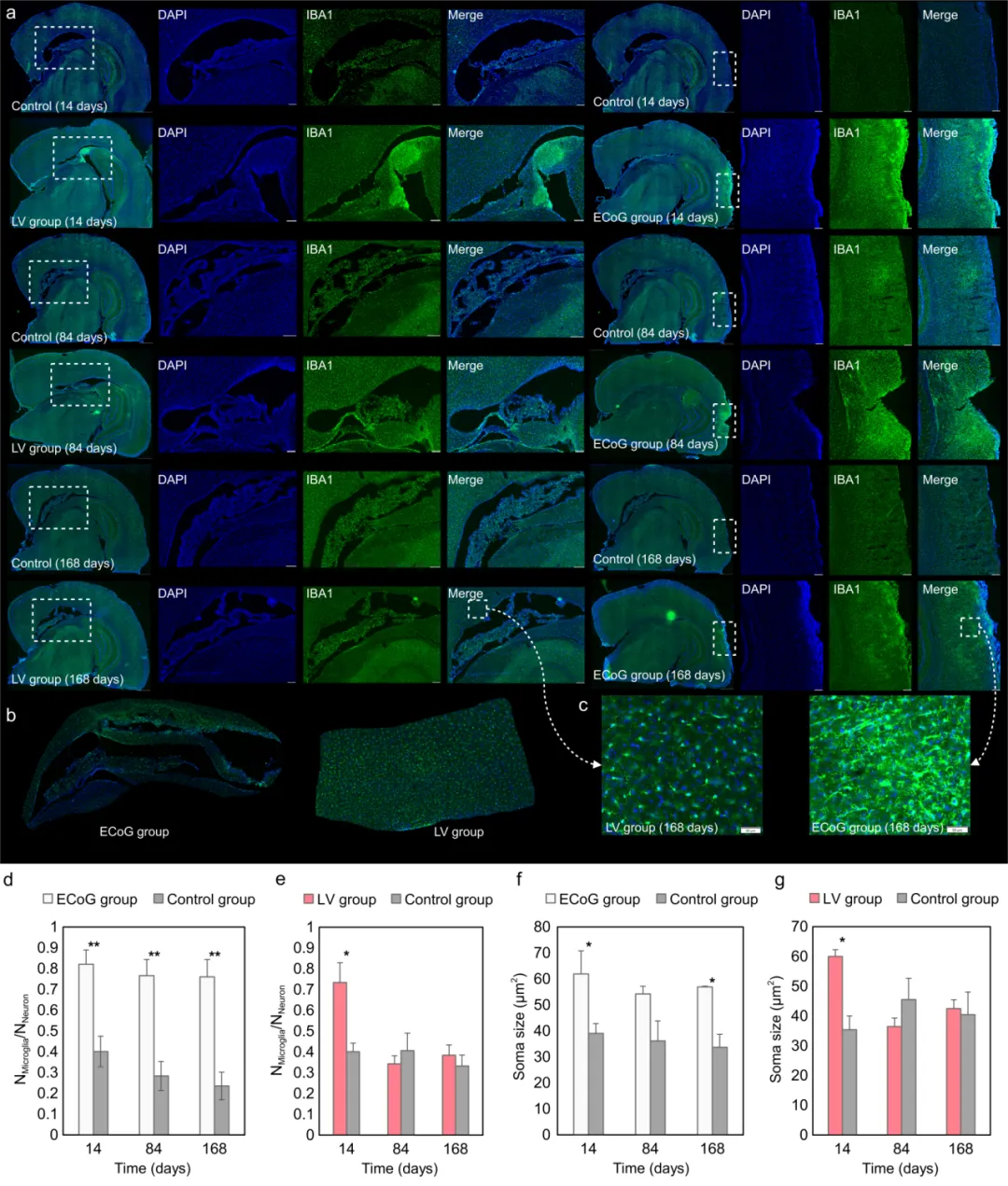
Comparison of immunostaining with traditional subdural ECoG electrodes.
At the functional level, the study further tested the ventricular BCI's performance in a memory-guided decision-making task. In a Action-memory T-maze, classification models built using signals from the ventricular electrode achieved significantly higher predictive accuracy for the animals' impending choices, peaking near 98%, overall outperforming cortical ECoG significantly. Feature analysis indicated that the discriminative information captured by the ventricular interface more prominently involved regions adjacent to the hippocampus and striatum, suggesting a potential advantage in accessing network activity related to memory and decision-making.
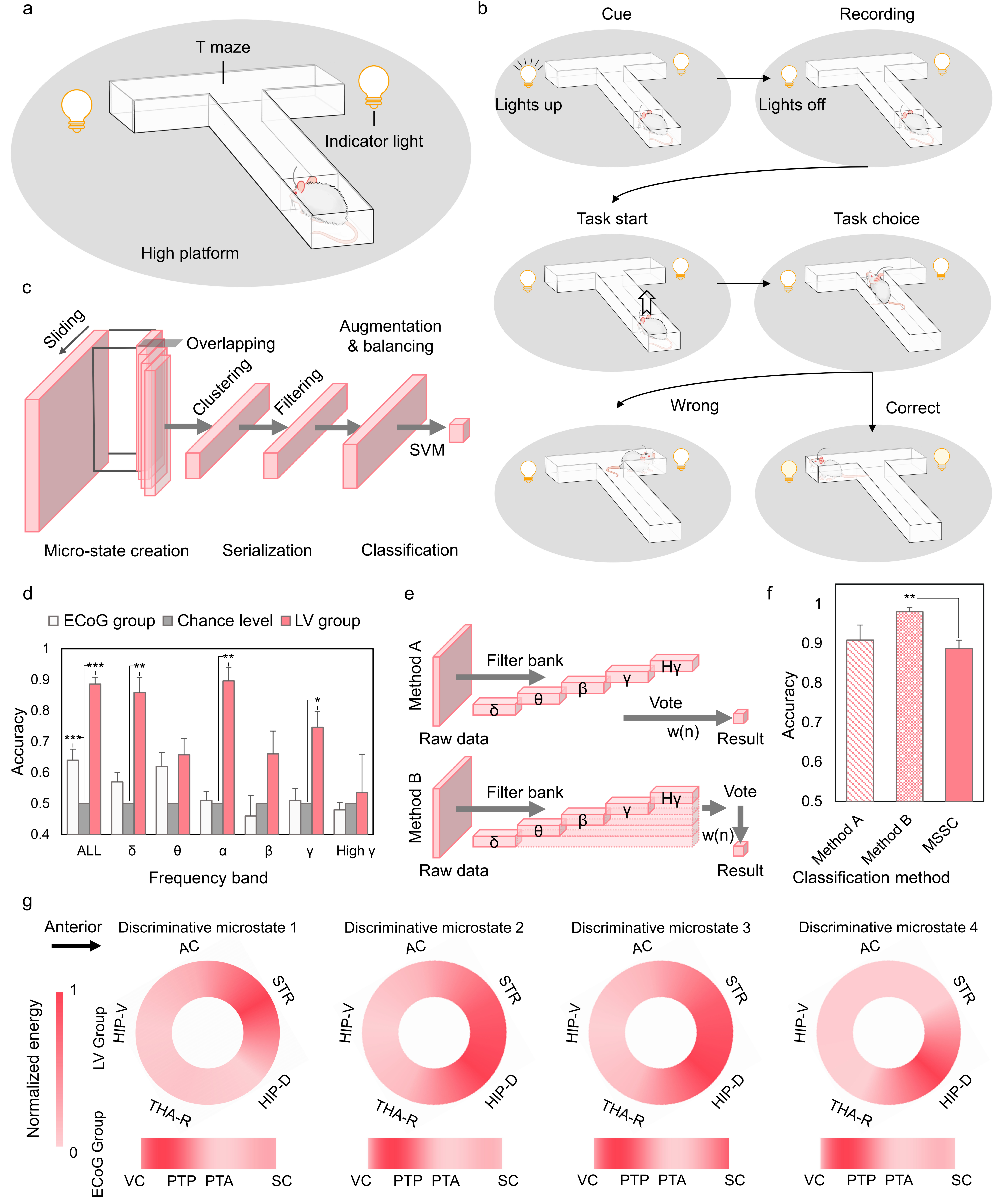
Memory classification task design and classification results
Overall, this study proposes a BCI approach distinct from traditional cortical pathways, innovating at both the electrode structural design and the implantation location selection levels. The application of the lantern-inspired expandable electrode within the ventricular environment offers a new technical direction for enhancing the long-term stability of invasive BCIs and opens new potential pathways for accessing information related to deep cognitive functions. Future work requires further validation regarding device scale optimization, adaptation to the human ventricular structure, and long-term safety assessment. Nevertheless, the proposed "ventricular pathway" concept provides a new reference framework for BCI design.
Dr. Yike Sun, a 2025 PhD graduate from the School of Biomedical Engineering, Tsinghua University, and currently an Assistant Professor at Beihang University; Yaxuan Gao, a PhD student at the School of Pharmaceutical Sciences, Tsinghua University; and Kewei Wang, a 2024 Master's graduate from the Biomedical Engineering program at Tsinghua Shenzhen International Graduate School, are the co-first authors of the paper. Professor Xiaorong Gao, Professor Ran Liu from the School of Biomedical Engineering, Tsinghua University, Professor Bai Lu from the School of Pharmaceutical Sciences, and Professor Xiaogang Chen from the Chinese Academy of Medical Sciences are the corresponding authors.


