Minimally invasive deep brain neuromodulation represents a long-standing key technological challenge in the field of neuroscience and neural engineering, holding significant importance for deciphering brain function and intervening in neurological disorders. Low-intensity focused ultrasound (LIFU) is considered a potentially vital technological means for achieving deep, wireless neuromodulation due to its excellent tissue penetration capability and spatial focusing characteristics. However, neuromodulation relying solely on ultrasound can yield responses that depend on neuronal subtype composition, mechanosensitive ion channel expression, and the local mechanical microenvironment, thereby contributing to variability in stimulation outcomes and affecting the stability and predictability of modulation. In recent years, functionalized nanoparticles have been explored as “acoustic–electrical” or “acoustic–mechanical” energy transduction units to facilitate more defined local energy conversion during ultrasound exposure. Therefore, developing nanoscale energy transduction and modulation techniques that require no transgenesis, no electrode implantation, and can achieve stable, predictable neural activation under low-intensity ultrasound conditions is of great significance for advancing the refinement and clinical translation of wireless neuromodulation technologies.
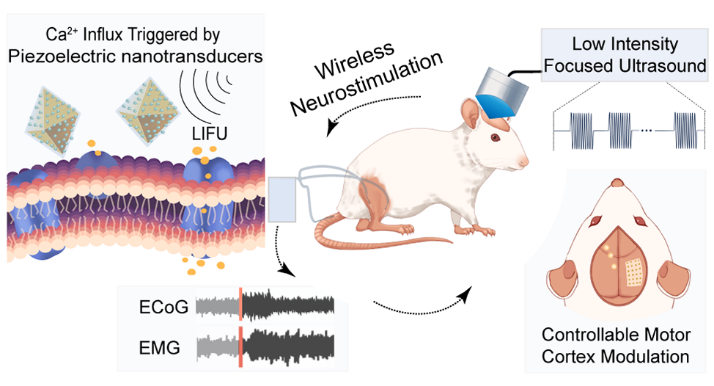
Figure 1. Schematic illustration of wireless ultrasound neuromodulation mediated by aggregation-enhanced piezoelectric nanotransducers.
Recently, the research teams led by Professor Mingjun Zhang from the School of Biomedical Engineering, Tsinghua University, and Professor Changxiang Yan from Sanbo Brain Hospital, Capital Medical University, investigated a method based on functionalized nanoparticles for aggregation-enhanced piezoelectric nanotransduction, enabling wireless neurostimulation under low-intensity focused ultrasound. The study, titled "Aggregation-Enhanced Piezoelectric Nanotransducers Facilitate Transgene-Free Wireless Neurostimulation under Low-Intensity Focused Ultrasound," was published in the journal ACS Nano. While low-intensity ultrasound itself possesses favorable tissue penetration and spatial focusing capabilities, positioning it as an important technological approach for deep brain neuromodulation, its neuromodulatory efficacy often suffers from uncertainties arising from variations in tissue and cellular characteristics. In this study, 10 nm barium titanate (BaTiO₃) piezoelectric nanoparticles were assembled onto the surface of porous UiO-66 metal-organic frameworks, followed by the introduction of a conductive polydopamine (pDA) coating. This structural configuration enhanced local electric field generation under ultrasound stimulation and reduced cellular internalization compared with dispersed nanoparticles. Experimental results demonstrated that this nanotransducer system could stably induce calcium ion influx mediated by voltage-gated ion channels in differentiated PC12 cells. Furthermore, upon injection into the rat motor cortex, it achieved spatially specific electrocorticographic responses and corresponding electromyographic outputs from relevant body parts upon LIFU stimulation. This structural configuration enhanced local electric field generation under ultrasound stimulation and reduced cellular internalization compared with dispersed nanoparticles. Collectively, these results demonstrate a nanoparticle-assisted ultrasound energy transduction strategy for wireless neuromodulation and provide a structural design framework for micro–nano transducers operating under low-intensity ultrasound.
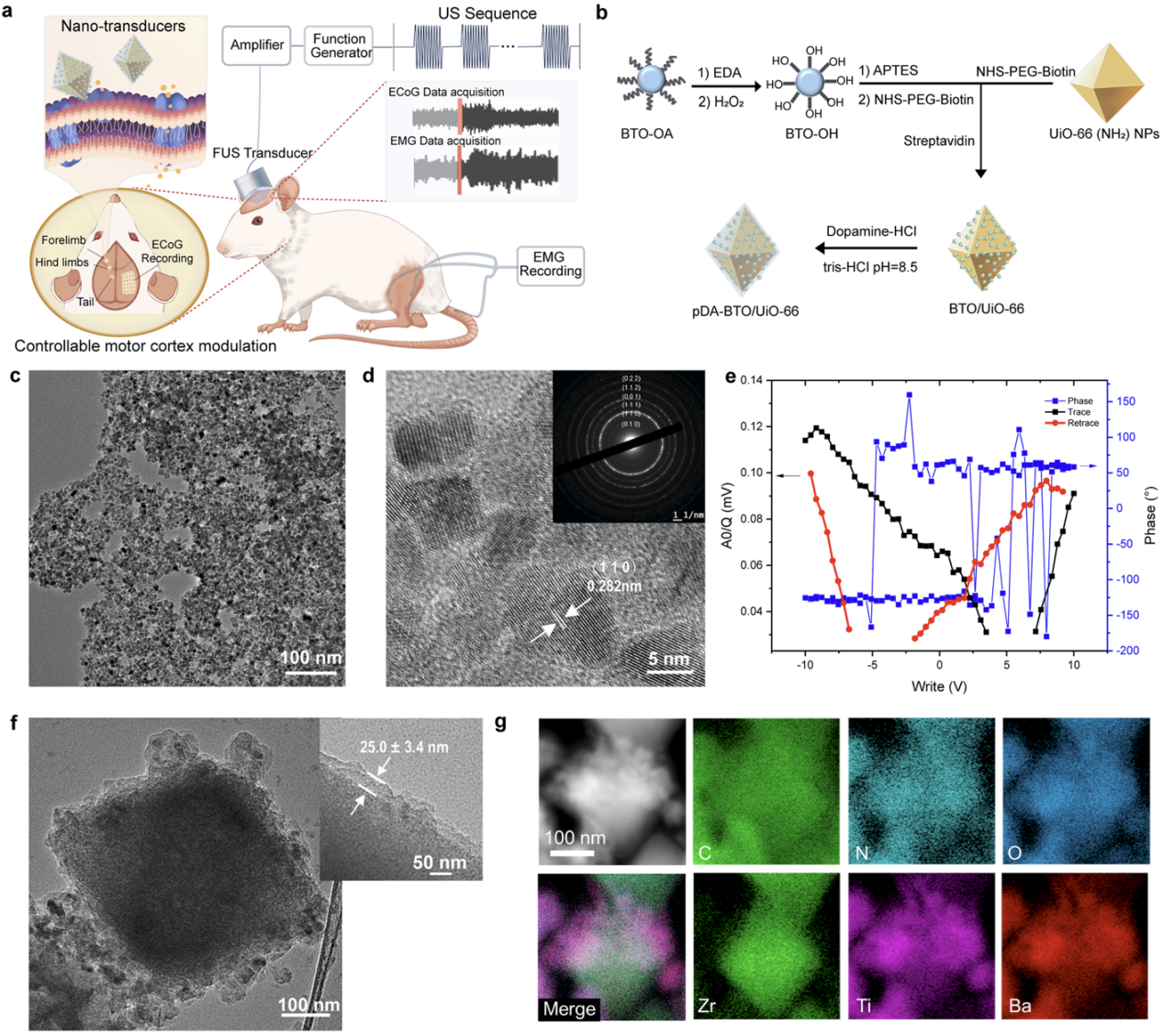
Figure 2. Schematic illustration of the research strategy for wireless neuromodulation using piezoelectric nanotransducers and corresponding material characterization.
To address the issues that dispersed individual piezoelectric nanoparticles generate weak local electric fields and are prone to cellular phagocytosis, hindering effective neuronal stimulation, this study proposed the design concept of "aggregation-enhanced" piezoelectric nanotransducers. The design utilizes UiO-66 metal-organic frameworks as the core, providing a high surface-area-to-volume ratio platform and abundant surface-active sites for loading the piezoelectric component. 10 nm barium titanate piezoelectric nanoparticles are densely assembled onto this surface, enabling a synergistic piezoelectric effect upon ultrasound stimulation to further enhance local electric field output. The outer layer incorporates a conductive polydopamine (pDA) coating to improve biocompatibility and colloidal stability, while further optimizing the coupling and transmission of the local electric field, thereby enhancing electrical conductivity. This structural design allows the nanotransducers to generate enhanced piezoelectric output under low-intensity ultrasound conditions and tend to adhere to the cell membrane surface, thus achieving sustained and effective membrane-associated electrical stimulation (Figure 3).
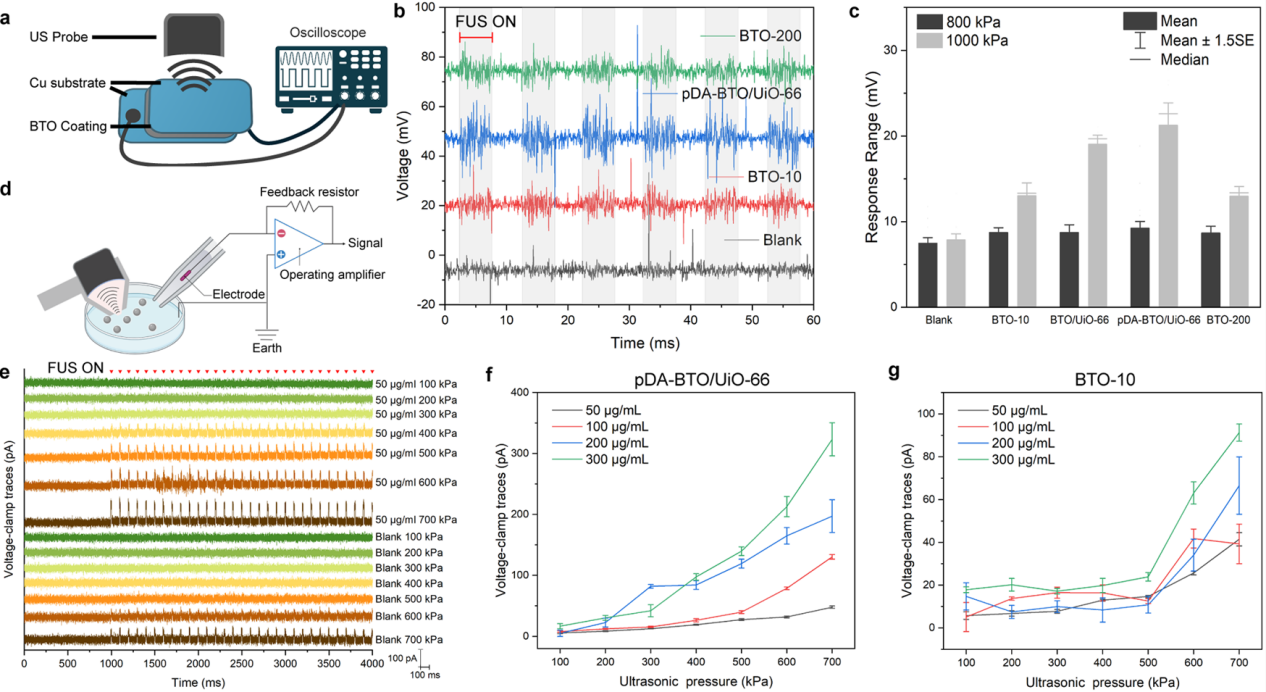
Figure 3. Response of piezoelectric nanotransducers under LIFU stimulation.
In a PC12 neuron-like cell model (Figure 4), this ultrasound-driven piezoelectric nanotransducer system could induce intracellular calcium ion influx in a tunable manner, demonstrating its effective neural activation capability. Further mechanistic studies revealed that this calcium response primarily depended on the influx of extracellular calcium ions and could be significantly blocked by voltage-gated sodium channel or calcium channel inhibitors, whereas inhibition of mechanosensitive channels had a limited effect. These results indicate that this design can induce membrane potential changes via the piezoelectric effect, activating voltage-gated ion channels to achieve neuromodulation, rather than relying on mechanical stimulation mechanisms.
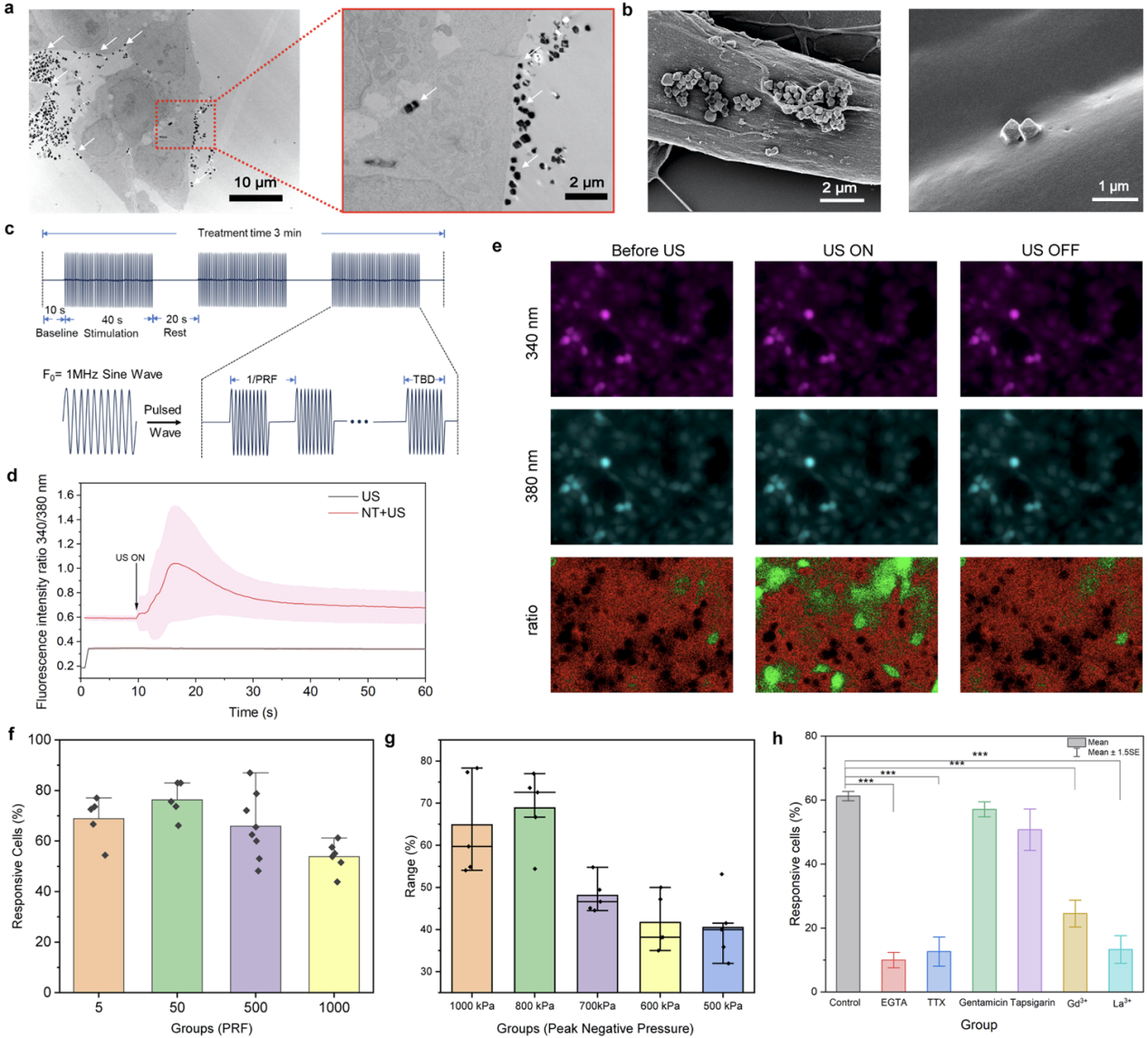
Figure 4. In vitro piezoelectric neurostimulation using nanotransducers and mechanistic investigation.
Following injection of the aforementioned piezoelectric nanotransducers into specific regions of the rat motor cortex and subsequent application of low-intensity focused ultrasound stimulation (Figure 5), robust and stable neural responses were observed in electrocorticography (ECoG) recordings. Simultaneously, electromyography (EMG) results indicated that stimulating different functional subregions of the motor cortex (corresponding to the forelimb, hindlimb, and tail, respectively) elicited muscle activity specific to the respective body parts, demonstrating the excellent spatial selectivity and functional specificity of this system. Further analysis via c-Fos immunofluorescence staining confirmed significant neuronal activation in the stimulated regions, with activation levels comparable to those achieved by invasive electrode stimulation, thereby validating the efficacy of this wireless neuromodulation strategy under in vivo conditions.
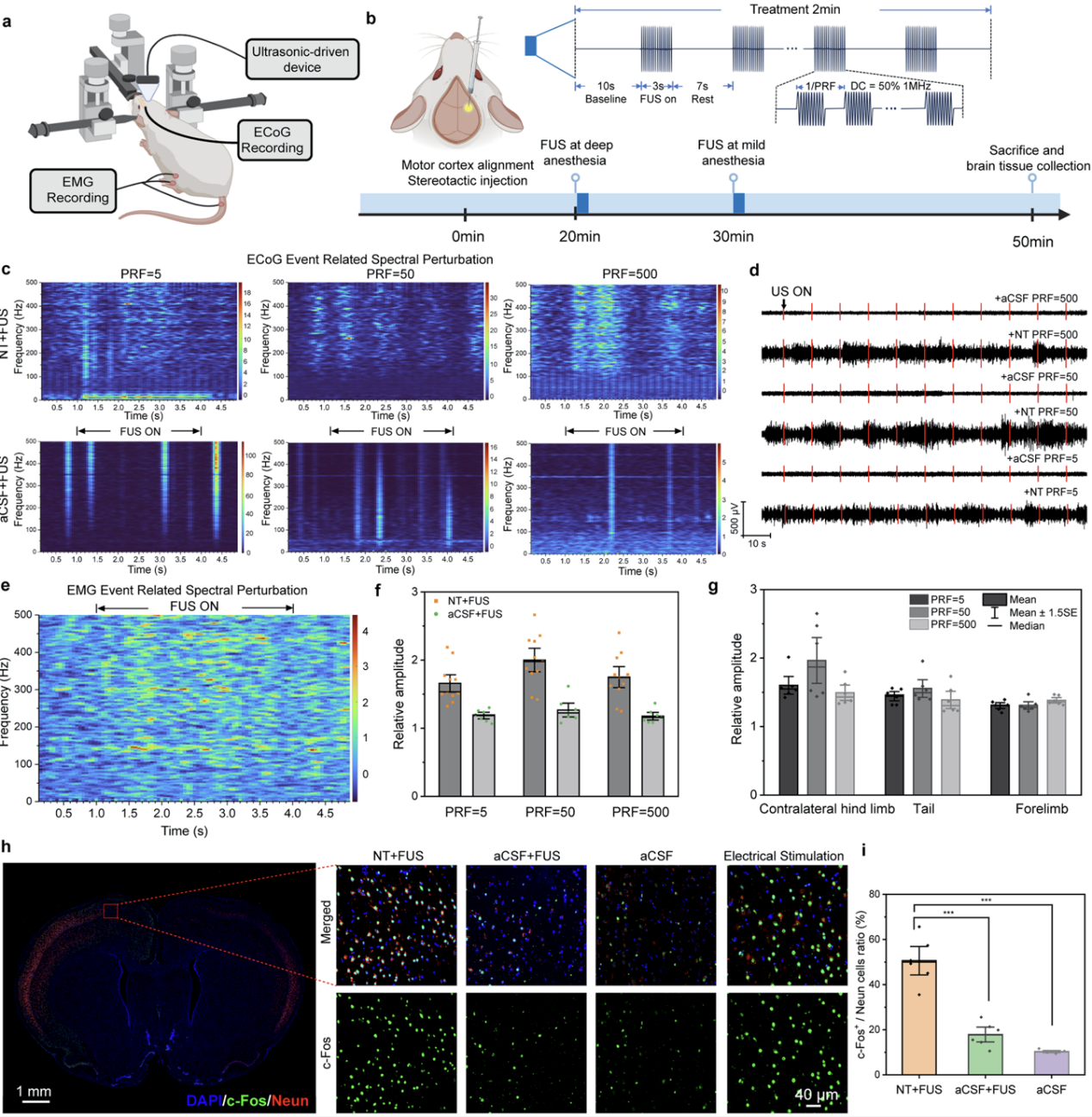
Figure 5. Multi-site piezoelectric neurostimulation in the rat motor cortex using nanotransducers.
Biosafety evaluation results (Figure 6) indicated that this nanotransducer system could be rapidly cleared from brain tissue. Short-term and long-term histological assessments revealed no neuronal loss, nor any increase in astrocyte or microglial density following treatment, suggesting its favorable biocompatibility and neural safety.
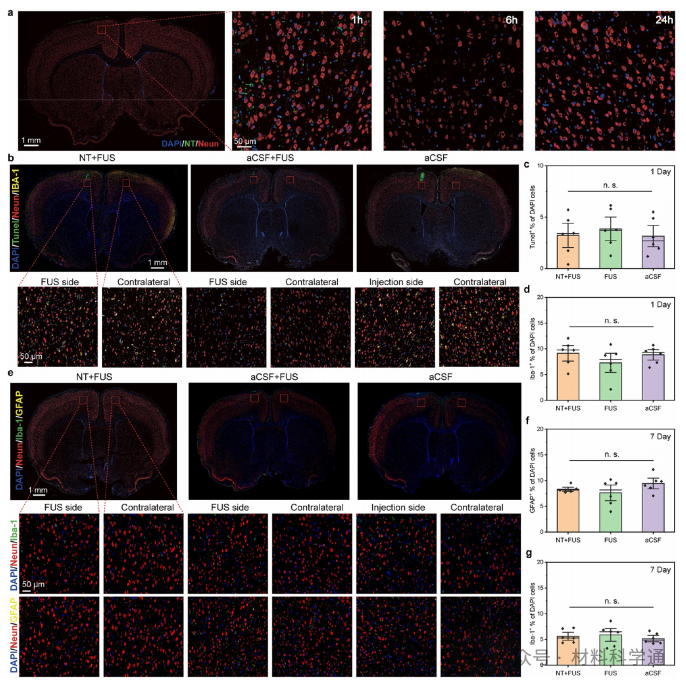
Figure 6. Biosafety evaluation.
This study presents a wireless neuromodulation paradigm requiring neither transgenesis nor electrode implantation. Low-intensity ultrasound functions as a deep-penetrating energy carrier, while functionalized piezoelectric nanoparticles serve as efficient energy transducers that convert acoustic input into localized electrical stimulation through rational micro–nano structural design. Mechanistically, this approach significantly reduces dependence on the heterogeneity of mechanosensitive ion channels. Looking ahead, combined with non-invasive delivery strategies such as reversible blood-brain barrier opening, this nanoparticle-assisted ultrasound neuromodulation system holds substantial potential translational potential for both fundamental neuroscience research and future clinical neuromodulation applications.
Professor Mingjun Zhang from the School of Biomedical Engineering, Tsinghua University, and Professor Changxiang Yan from Sanbo Brain Hospital, Capital Medical University, are the co-corresponding authors. Doctoral student Chengqian Cui (class of 2023) and Postdoctoral fellow (Ph.D. graduate of 2025) Xize Gao from the School of Biomedical Engineering, Tsinghua University, are the co-first authors. Doctoral students Nianzhen Du (class of 2022) and Jingwei Li (class of 2024) from the School of Biomedical Engineering, Tsinghua University, made significant contributions to this research.
Original Article Link:
https://doi.org/10.1021/acsnano.5c15915


