Background: The "Miniaturization" Challenge in Neuromodulation
In the frontier of brain-computer interfaces (BCIs) and neuroscience, achieving subcellular-scale neuromodulation is a critical challenge for decoding the computational principles of neural circuits. Neuronal information processing is highly localized within microscopic membrane domains, such as the soma and dendritic spines, typically spanning just a few micrometers. However, existing extracellular stimulation tools face a fundamental electrochemical trade-off when scaled down to subcellular scales (< 10 μm). As the geometric surface area drastically decreases, the interfacial impedance surges, resulting in insufficient charge-delivery capacity for effective neural activation.
For a long time, fabricating high-performance conductive polymer (e.g., PEDOT:PSS) coatings on high-density microelectrode arrays has been hindered by microscopic electrochemical kinetics. Traditional electrodeposition processes are highly susceptible to inhomogeneous local electric fields and mass transport fluctuations. At extreme miniaturization, this manifests as a severe "edge effect," causing irregular polymer overgrowth at electrode peripheries and even electrical shorting between adjacent sites.
Breakthrough: Harnessing Diffusion Rather Than Fighting It
Moving away from the conventional mindset of "fighting" non-uniform deposition, Associate Professor Xiaochuan Dai’s team from the School of Biomedical Engineering at Tsinghua University drew inspiration from classical steady-state diffusion theory in electrochemistry. The team recognized that electrodeposition on microelectrodes is fundamentally a highly confined, diffusion-controlled process. By precisely calculating the steady-state limiting current (Iss) for ultramicroelectrodes, the team innovatively proposed a Diffusion-Tuned Galvanostatic Deposition strategy. The core of this approach lies in working within physical constraints: by strictly maintaining the deposition current below the theoretical diffusion limit, the monomer consumption rate perfectly balances with the diffusion replenishment. This establishes a stable and independent mass transport environment for each microelectrode. This method not only eliminates edge-biased overgrowth but also achieves highly uniform modification across entire arrays, providing a scalable technological framework for the batch modification of high-density subcellular-scale electrode arrays. The research, titled "Subcellular-Scale Stimulation Electrode Arrays (3SEA) Enabled by Diffusion-Tuned PEDOT:PSS Galvanostatic Deposition," was published online in the prestigious journal Nano Letters on March 3, 2026.

Click to view more paper information
To overcome the spatial limitations of neural interfaces, the team developed Subcellular-Scale Stimulation Electrode Arrays (3SEA). Figure 1 illustrates the comprehensive workflow, from the diffusion-tuned electrodeposition process to single-cell-level neural stimulation. Compared to existing silicon-based rigid electrodes, flexible thin-film electrodes, and traditional conductive polymer-coated electrodes, 3SEA maintains subcellular dimensions (3–10 μm) while significantly reducing interfacial impedance. This successfully breaks the inherent trade-off between miniaturization and electrochemical performance, establishing a physical foundation for high-density neural interfaces.
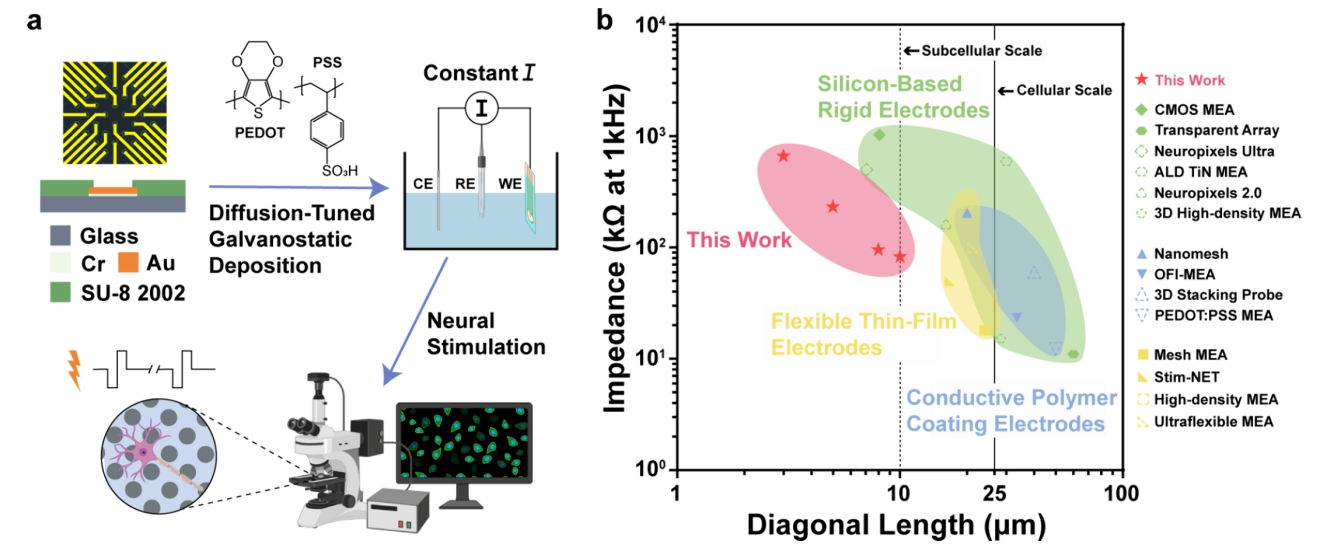
Figure 1: Technical roadmap of 3SEA and comparison of electrochemical performance with various representative neural electrodes.
By establishing a steady-state diffusion model for ultramicroelectrodes, the research team calculated the limiting current (Iss) for electrodes of different sizes to guide galvanostatic deposition parameters. Experiments confirmed that when the current per channel is controlled below Iss, the potential stabilizes within the optimal polymerization window, yielding a uniform PEDOT:PSS morphology without edge overgrowth. In contrast, conventional potentiostatic deposition leads to severe edge-biased nucleation. This physics-constraint regulation ensures consistency in impedance distribution across multi-channel arrays.
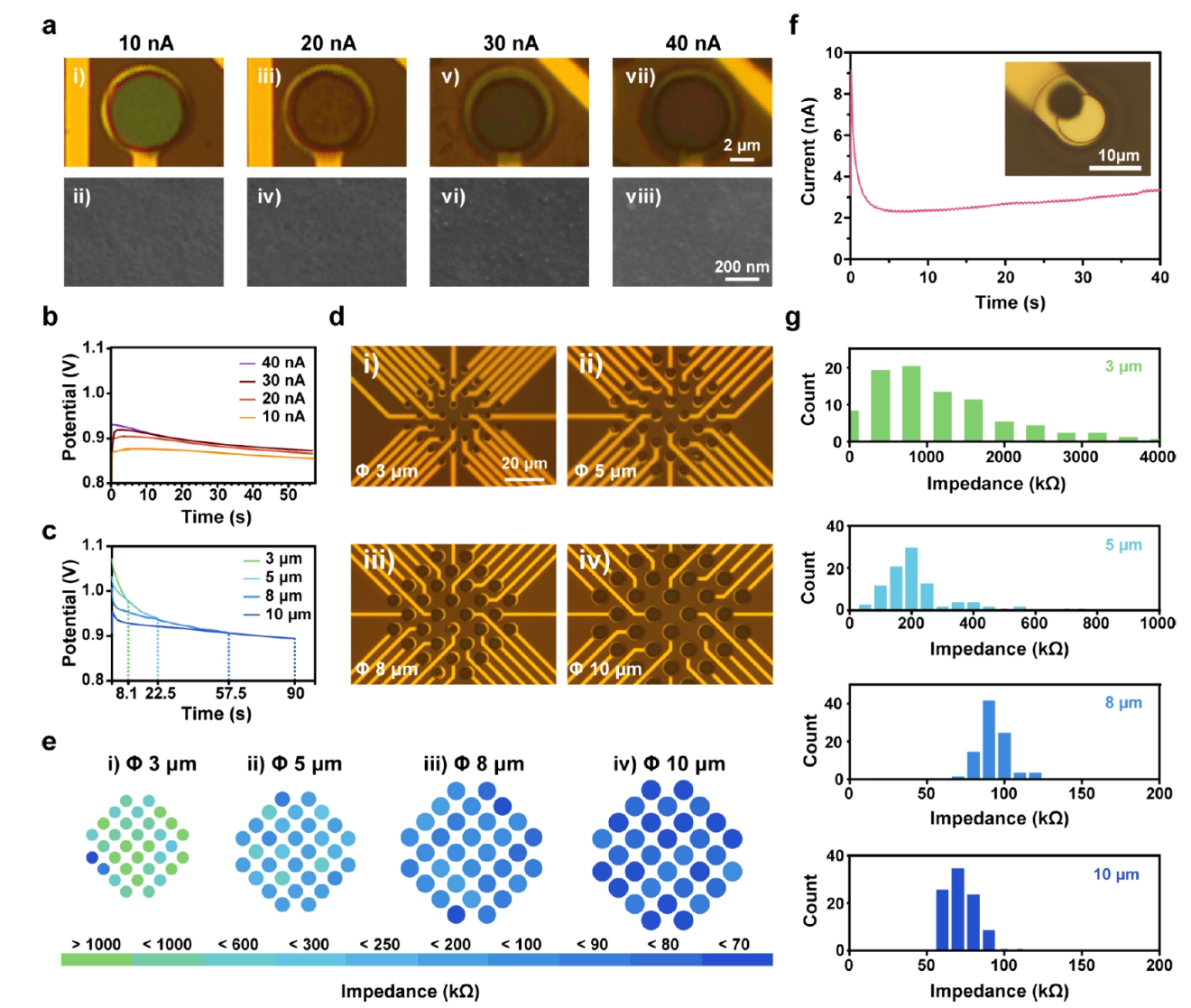
Figure 2: Optimization of uniform morphology and impedance consistency of 3SEA via the diffusion-tuned strategy.
The introduction of PEDOT:PSS not only reduces interfacial impedance by 6- to 30-fold but also significantly enhances the charge storage capacity (CSC) and charge injection capacity (CIC) by constructing a highly porous, mixed ionic-electronic conductive network. This robust charge-delivery capability, sustained even at subcellular effective areas, provides essential technological support for safe and efficient neural stimulation.
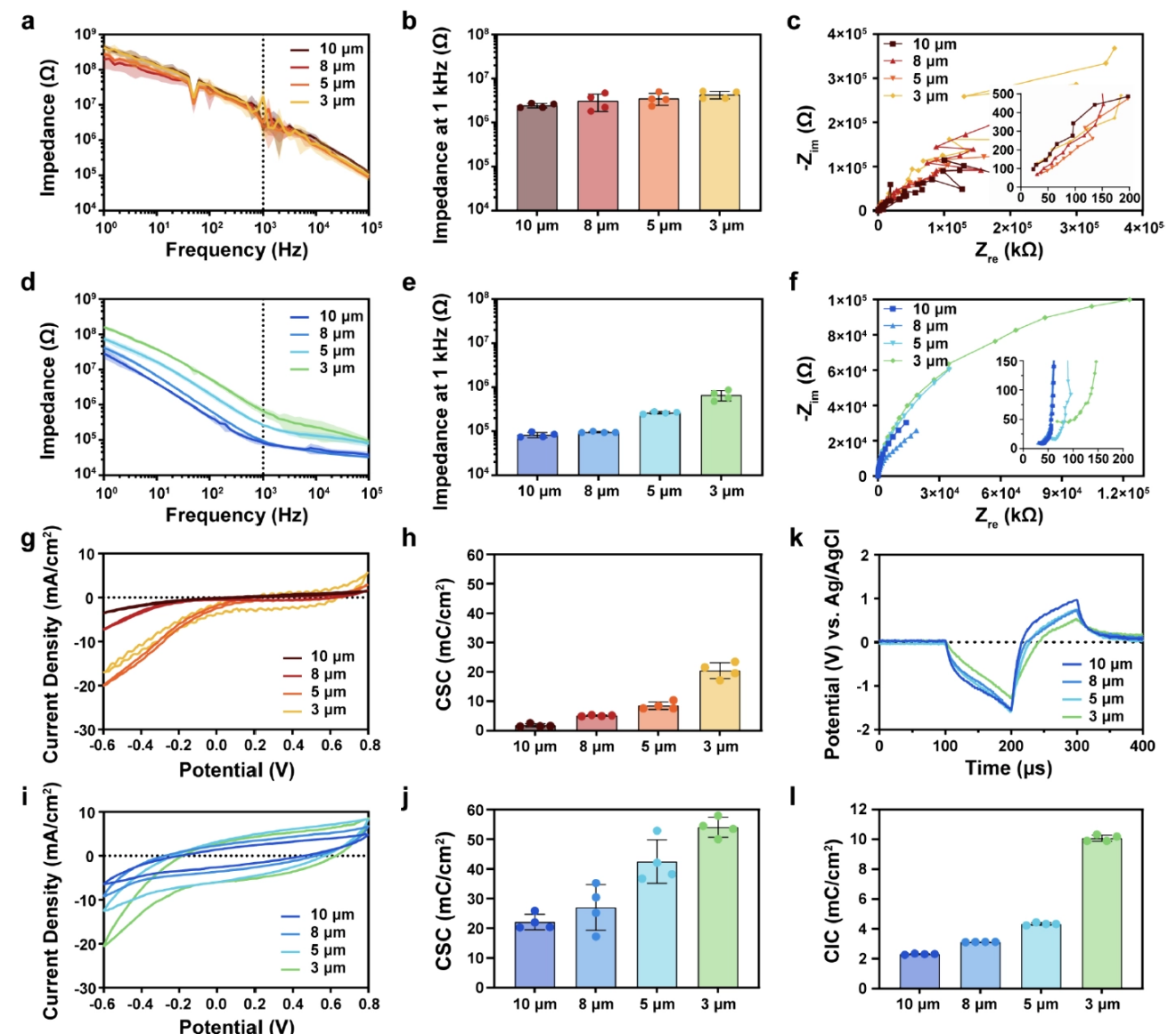
Figure 3: Electrochemical performance comparison before and after modification, including electrochemical impedance spectroscopy (EIS), cyclic voltammetry (CV), and voltage transients.
Functional Validation: Precise Evocation of Single-Cell Calcium Responses
The research team further cultured HT-22 cells on the 3SEA arrays. Fluorescence imaging visually demonstrated the physical size comparison between electrodes and neuronal microstructures: the 10 μm electrodes are slightly smaller than the soma, while 3–5 μm sites can precisely target finer subcellular structures like synapses. Functional neural stimulation experiments proved that 3SEA requires only a low charge of 1 nC/phase in biphasic pulses to precisely evoke intracellular calcium transients in target neurons. Spatiotemporal dynamics analysis revealed that the calcium signals originated strictly at the electrode-membrane interface (ROI) before propagating throughout the soma. In contrast, unmodified electrodes driven with the same charge rapidly reached water electrolysis thresholds, generating reactive species and causing cellular damage, further highlighting the biosafety and stimulation efficiency of the 3SEA technology.
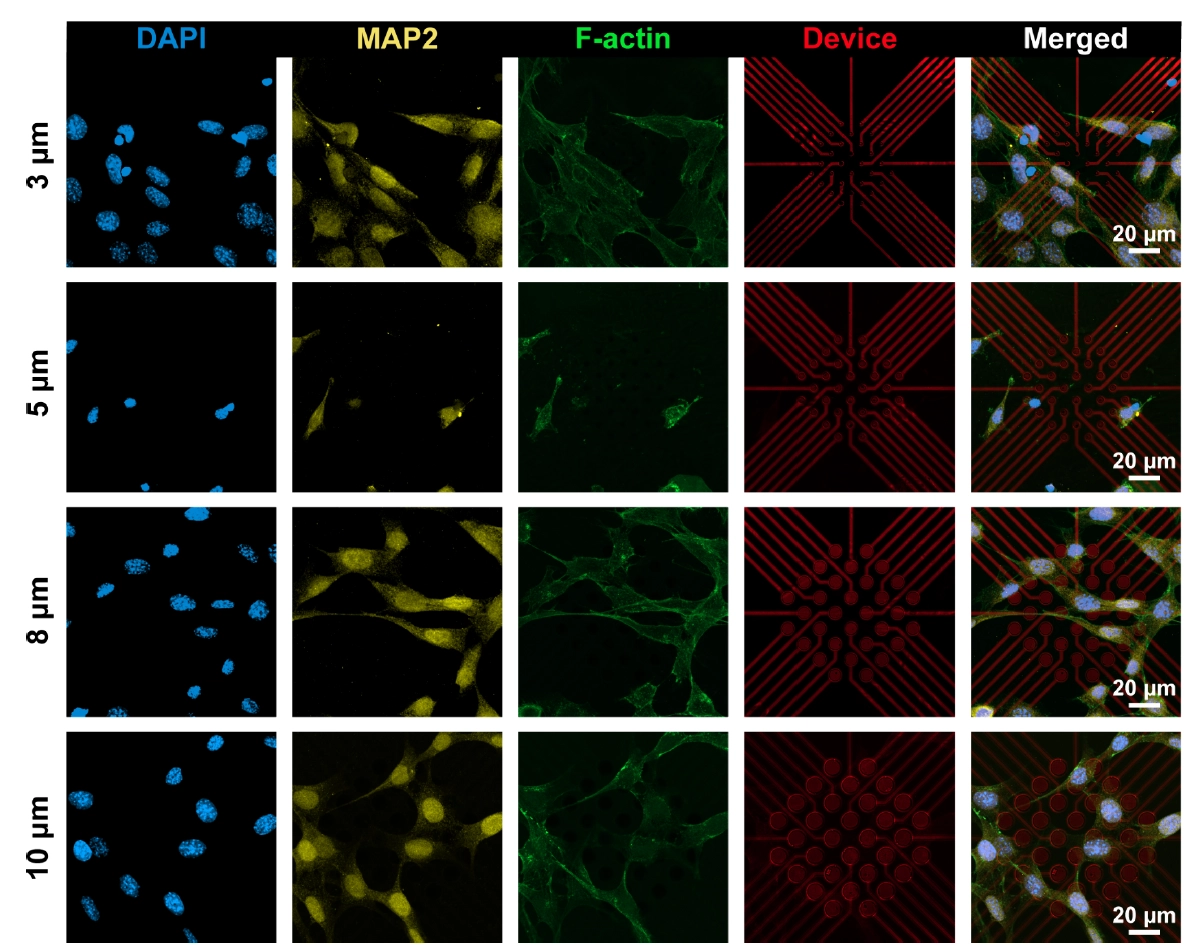
Figure 4: Fluorescence microscopy images of HT-22 cells adhering to 3SEA, illustrating the subcellular-scale size comparison.
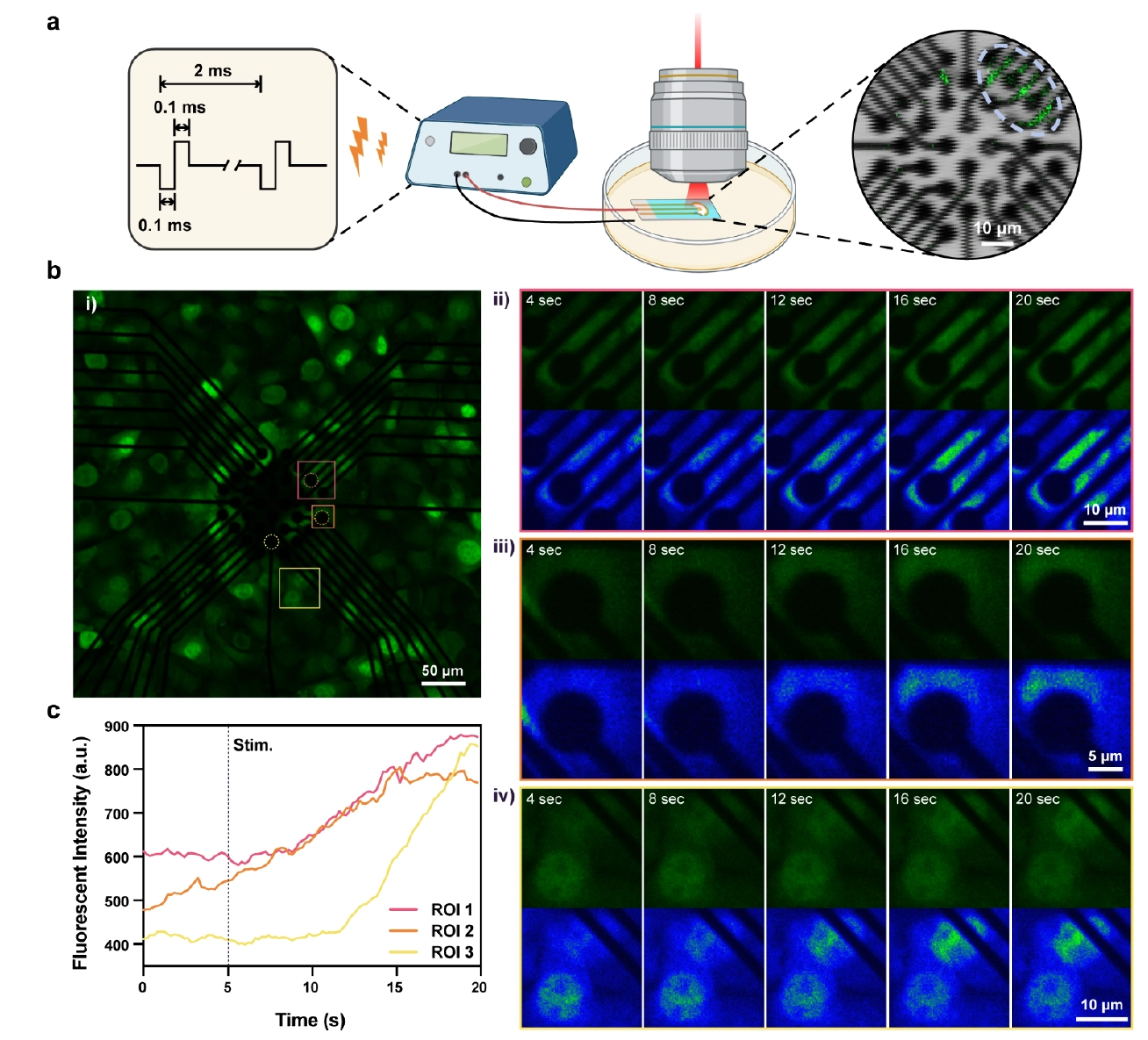
Figure 5: Cellular response to electrical stimulation via 3SEA, showing precise subcellular calcium signal triggering and its spatiotemporal dynamics.
Summary and Outlook
By introducing the Diffusion-Tuned Galvanostatic Deposition strategy, this study achieves, for the first time, uniform and reliable modification of subcellular-scale (3–10 μm) electrodes on high-density arrays. The 3SEA technology not only overcomes the electrochemical performance bottlenecks inherent in ultramicroelectrode miniaturization, but also demonstrates the potential for precise, subcellular-level neuromodulation. This paradigm shift from "fighting diffusion" to "harnessing diffusion" leverages physical constraints to achieve precise modifications, offering a versatile scientific framework for the tailored fabrication of high-performance, high-density bioelectronic devices.
Looking forward, 3SEA technology presents broad application potential:
Generalizability for Applications: This diffusion-tuned strategy can be readily adapted to flexible substrates and three-dimensional electrode architectures, paving the way for high-performance, minimally invasive in vivo neural probes.
Deciphering Single-Neuron Computational Dynamics: The capability for subcellular-scale stimulation empowers researchers to investigate intricate information processing mechanisms within individual neurons, such as dendritic computations and the dynamics of the axon initial segment.
Next-Generation Brain-Computer Interfaces and Precision Medicine: The high-density integration of 3SEA makes it a promising candidate for high-resolution retinal prosthetics, peripheral nerve interfaces, and closed-loop neural prosthetic systems, ultimately enhancing therapeutic efficacy through precise modulation.
Read the full paper: https://pubs.acs.org/doi/10.1021/acs.nanolett.5c06350



