For patients with heart disease, implantable cardiac pacemakers are "life-saving devices" that restore normal heart rhythm. By precisely regulating cellular activity, implantable bioelectronic devices not only restore cardiac function but are also widely used in areas such as motor function restoration, vision and hearing recovery, pain management, and disease diagnosis. They provide critical support for early intervention, precision treatment, and long-term management of major diseases, significantly improving patients' quality of life and reducing the burden on family caregivers. However, the depletion of device batteries necessitates secondary replacement surgeries, posing a significant challenge. Compared to the initial implantation, secondary surgeries involve greater trauma, higher risks, and impose a substantial economic burden. Therefore, "lifelong maintenance-free operation" has become the ultimate goal in this field, and the key to achieving this goal lies in overcoming the technical bottleneck of lifelong energy supply.
Conceptual Innovation
From "Passive Energy Consumption" to "Active Energy Supply"
To address this challenge, Professor Li Zhou's team at the School of Biomedical Engineering, Tsinghua University, moved beyond traditional device design paradigms. They broke through the conventional assumption that electronic devices must rely on batteries or wireless power, proposing to achieve energy supply for implantable electronic devices through human-body self-powering. The research team developed the first self-powered cardiac pacemaker in 2014 (Advanced Materials, 2014, 26(33):5851-5856). In 2019, they successfully developed a symbiotic self-powered cardiac pacemaker, with related results published in Nature Communications (2019, 10, 1821), introducing the innovative concept of "symbiotic bioelectronics." Subsequently, the team has continuously expanded the application boundaries of these self-powered bioelectronic devices, achieving significant progress in fields such as bone repair, neural modulation, and bioresorbable electronics (Nature Communications, 2024, 15:507; Science Advances, 2024, eadi6799; Device, 2025, 3, 100724; Advanced Science, 2024, 2410289).
Technological Breakthrough
Collaborative Effort Creates "Human-Machine Symbiotic" Miniaturized Self-Powered Pacemaker
After nearly seven years of collaborative research, Professor Li Zhou and his partners, Professor Hua Wei from Fuwai Hospital, Chinese Academy of Medical Sciences, and Associate Professor Ouyang Han from the University of Chinese Academy of Sciences, along with their research team, have finally achieved a significant breakthrough—successfully developing a capsule-sized micro symbiotic self-powered leadless cardiac pacemaker. The related research findings, titled "Symbiotic transcatheter pacemaker for lifelong energy regeneration and therapeutic function in porcine disease model," were published in Nature Biomedical Engineering on January 19, 2026.

Click for more information on the paper
The core innovation of this pacemaker lies in the integration of a highly efficient energy regeneration module: It harnesses kinetic energy from the heart's own beating via electromagnetic induction technology and converts it into electrical energy. Tests show its output power has exceeded the critical energy threshold required for lifelong pacemaker operation, enabling it to stably drive pacing circuitry and achieve precise regulation of cardiac rhythm. Simultaneously, the device features a highly miniaturized design, possesses excellent biocompatibility and hemocompatibility, and supports transcatheter minimally invasive implantation, significantly reducing surgical trauma. The team innovatively designed an ultra-simplified magnetic levitation energy buffering structure, which not only minimizes energy loss and mechanical friction but also achieves an ultra-low startup threshold, high kinetic energy conversion efficiency, and stable average intracardiac output power, while simplifying system complexity and enhancing the device's long-term stability.
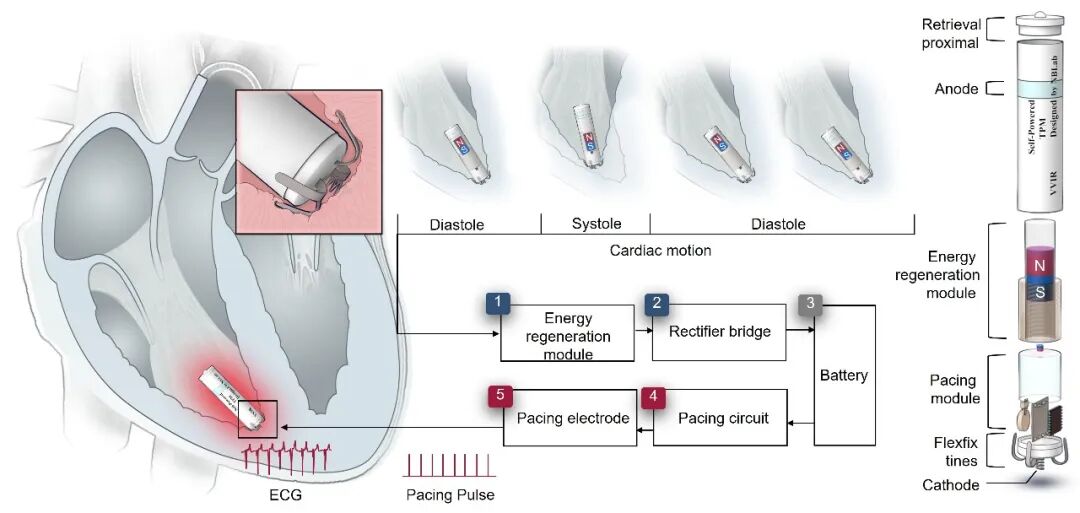
Schematic diagram of the symbiotic self-powered leadless cardiac pacemaker
Preclinical Evaluation
One-Month Long-Term Animal Study Tests Translational Potential
In a porcine model of third-degree atrioventricular block arrhythmia, this symbiotic pacemaker completed a one-month autonomous operation test. Throughout the experiment, it continuously achieved self-sustaining energy supply while stably performing pacing therapy functions, effectively regulating the heart rhythm of the experimental animals, thus fully validating its potential for clinical translation.
This technological breakthrough holds the potential to extend the operational lifespan of pacemakers to match that of the natural heart, completely resolving the issue of secondary surgeries and opening a new pathway towards truly "lifelong maintenance-free" and "human-machine symbiotic" implantable electronic devices.
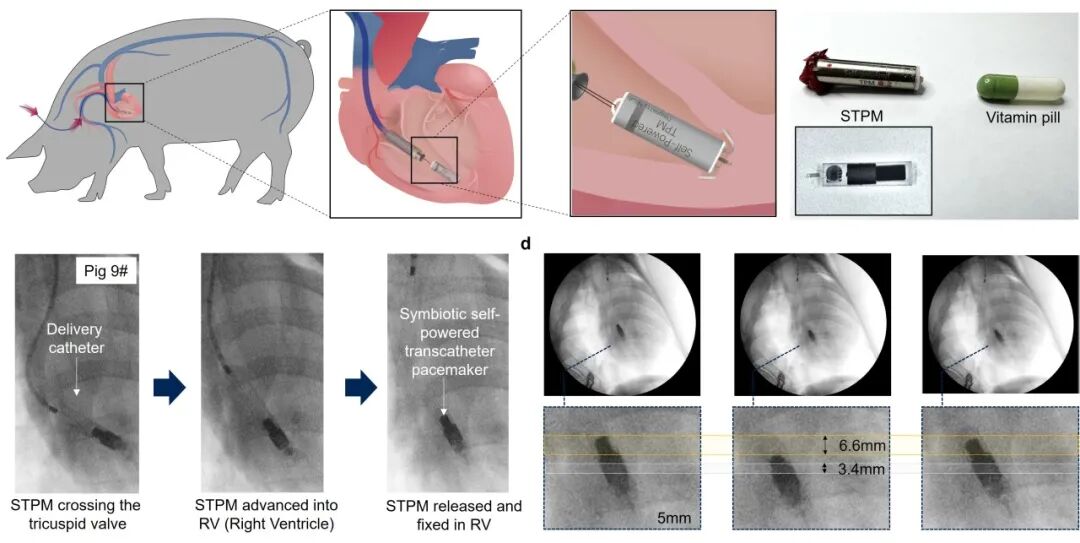
Diagram of the in-vivo implantation process and DR image
Professor Li Zhou from the School of Biomedical Engineering, Tsinghua University, and Beijing Tsinghua Changgung Hospital, and Professor Hua Wei from Fuwai Hospital, Chinese Academy of Medical Sciences, are the corresponding authors of this paper. Associate Professor Ouyang Han from the University of Chinese Academy of Sciences, Associate Professor Jiang Dongjie from Peking University/Peking University Third Hospital, Dr. Hu Yiran and Dr. Cheng Sijing from Fuwai Hospital, Chinese Academy of Medical Sciences, and Associate Professor Zhang Zhengmin from Hangzhou Dianzi University are the co-first authors.
Academician Wang Zhonglin from the Beijing Institute of Nanoenergy and Nanosystems, Chinese Academy of Sciences, provided significant support for this work. This research was jointly funded by the National Natural Science Foundation of China, the National Key Research and Development Program of China, the Youth Innovation Promotion Association of the Chinese Academy of Sciences, the Beijing Natural Science Foundation, the Beijing Nova Program, the Xinxi Disruptive Technology Innovation Fund, and the Fundamental Research Funds for the Central Universities.


