Cancer metastasis is a complex, multi-stage process, in which the presence of Circulating Tumor Cells (CTCs) serves as a crucial marker of tumor metastasis. Recent studies have revealed that intratumoral microbiota (such as bacteria) can alter the biophysical properties of host cells by influencing cytoskeletal reorganization, thereby enhancing the metastatic potential of tumor cells. However, there is currently a lack of efficient, high-throughput methods to detect these bacteria-infected CTCs with metastatic potential, posing a significant challenge for early prediction of cancer metastasis and therapeutic intervention.
Recently, a team led by Professor Hu Tony Ye from the School of Biomedical Engineering, Tsinghua University, and Professor Song Yujun from Nanjing University, developed a microfluidic-based cellular mechanical phenotype screening chip (LesM). This chip is designed to efficiently capture CTCs and assess the single-cell deformability changes induced by bacterial infection, thereby revealing their metastatic potential during hematogenous dissemination.The chip utilizes L-shaped trap structures to differentiate bacteria-infected CTCs from uninfected cells based on their deformability, enabling high-throughput detection of CTCs and metastasis risk assessment. This provides a novel tool for predicting distant metastasis in breast cancer and guiding antibacterial therapy. The research findings, titled "A Mechanophenotyping chip for high-throughput detection of metastatic bacteria-infected circulating tumor cells," were published in the journal (Nature Communications) on January 8, 2026.

Click for more information on the paper
Research Innovations
The research team designed a novel microfluidic chip, LesM, capable of efficiently capturing CTCs based on their mechanical phenotype and distinguishing between bacteria-infected and uninfected cells. They proposed a new method to assess the metastatic potential of CTCs through cell deformability, achieving high-sensitivity detection of bacteria-infected CTCs by combining fluorescence labeling techniques. In vivo experiments validated the correlation between chip detection results and tumor metastasis, and the impact of antibiotic treatment on tumor metastasis was explored, providing a theoretical basis for clinical application.
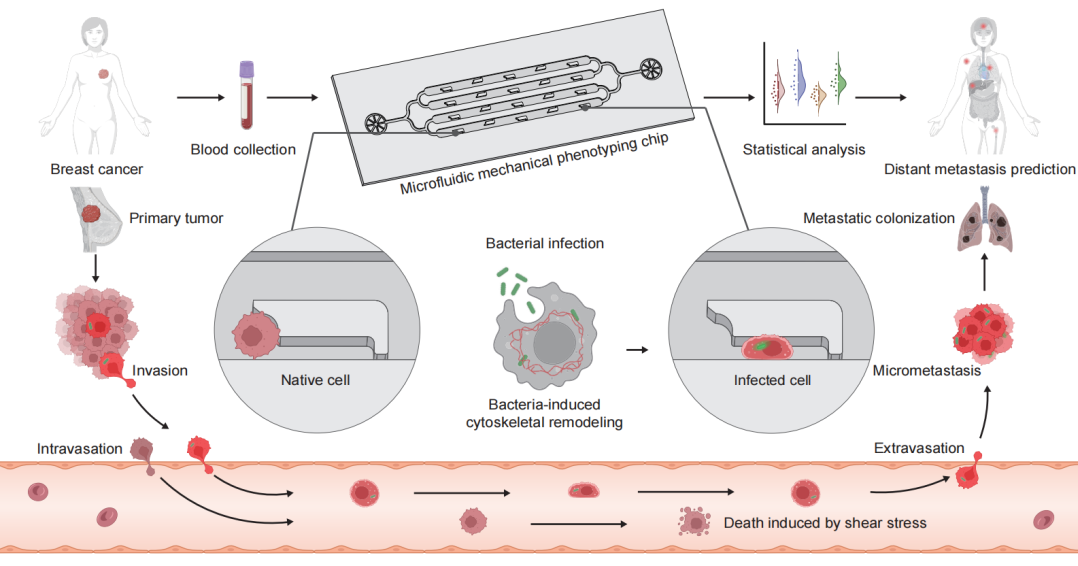
Figure 1: Design principle and workflow of the LesM chip.
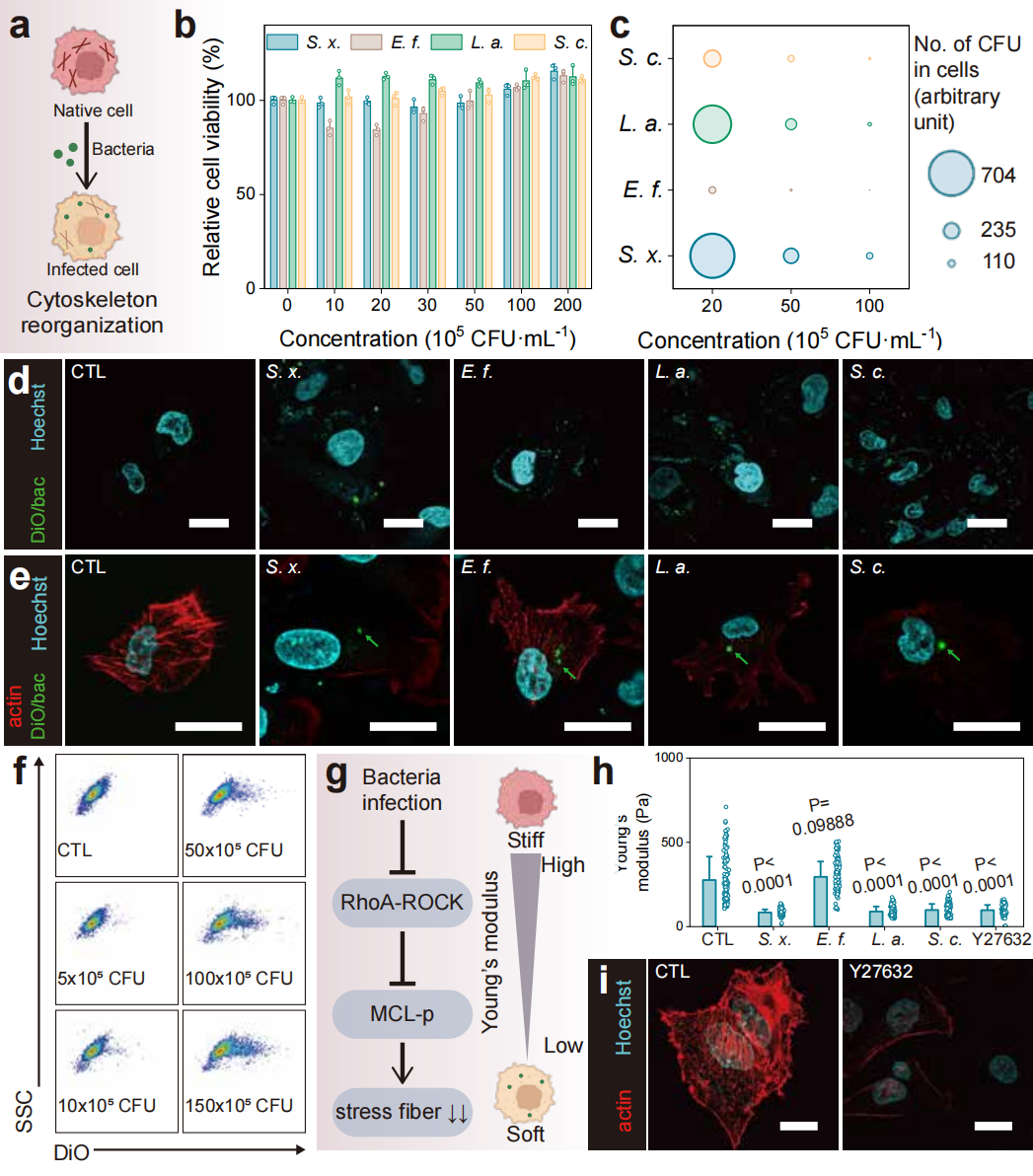
Figure 2: Impact of bacterial infection on the biophysical properties of breast cancer cells.
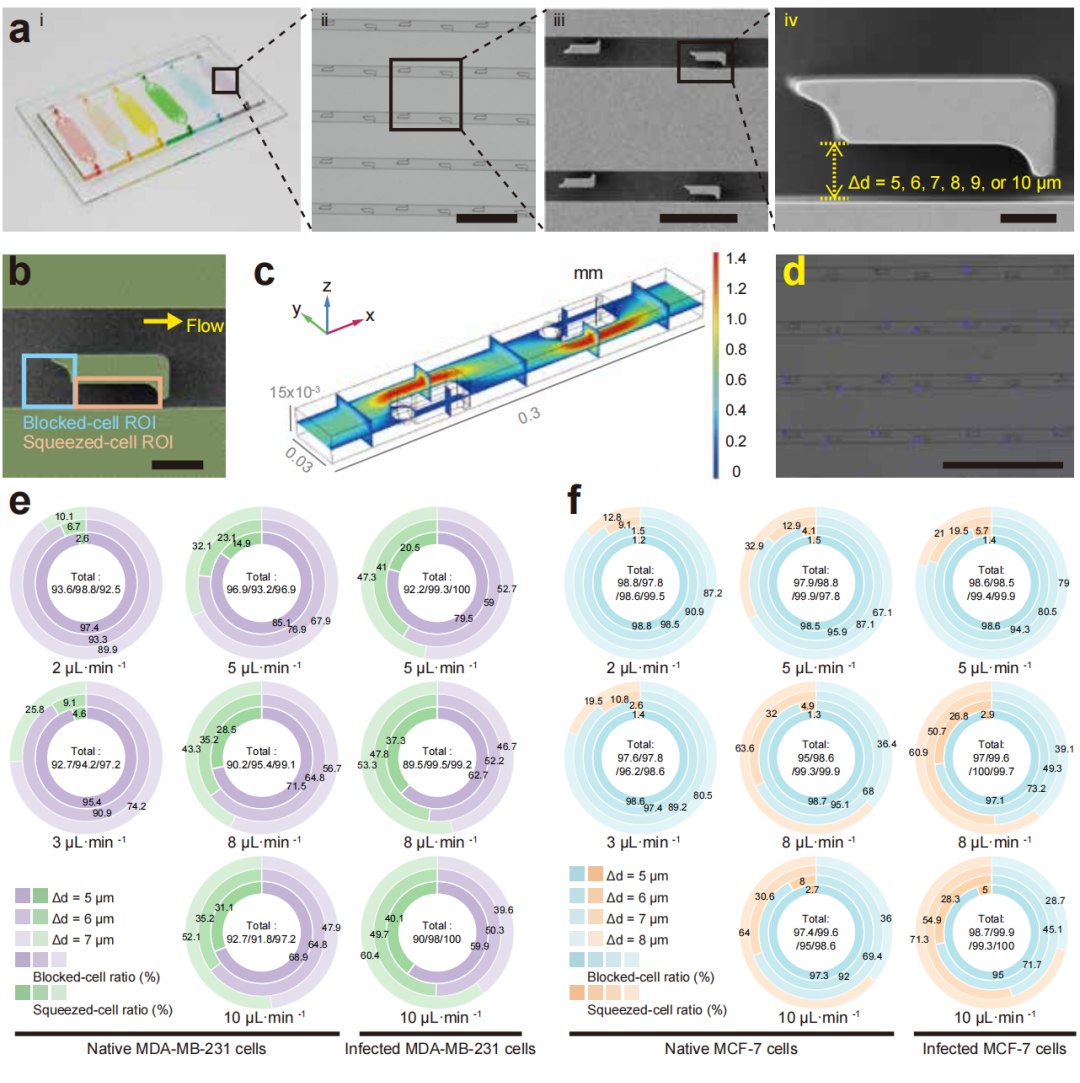
Figure 3: Structural design, optimization process, and evaluation of single-cell capture efficiency of the LesM chip.
Experimental Validation: Comprehensive Breakthrough from Cell Models to Clinical Samples
The research team thoroughly validated the performance and reliability of the LesM chip through a series of in vitro and in vivo experiments. Regarding cell-level validation, in both MDA-MB-231 and MCF-7 breast cancer cell models, it was demonstrated that bacteria such as Staphylococcus xylosus and Limosilactobacillus animalis could dismantle stress fibers by inhibiting the RhoA-ROCK signaling pathway, reducing the Young's modulus of the cells, and enhancing deformability. For simulated clinical sample detection, in experiments where healthy human blood was spiked with infected tumor cells, the chip efficiently distinguished tumor cells from blood cells. Using dual indicators of fluorescence intensity and cell size, it achieved high-sensitivity screening of infected circulating tumor cells. For animal model validation, in a mouse xenograft model of breast cancer, the number of infected circulating tumor cells detected by the LesM chip showed a significant positive correlation with the formation of lung metastatic foci. Moreover, pre-surgical antibiotic treatment to clear tumor-resident bacteria significantly reduced the number of infected circulating tumor cells and lowered metastasis risk. For clinical sample validation, analysis of whole blood samples from breast cancer patients revealed a 100% detection rate of infected circulating tumor cells in the distant metastasis group, with cell counts significantly higher than the other two groups, confirming the chip's predictive value for distant metastasis.
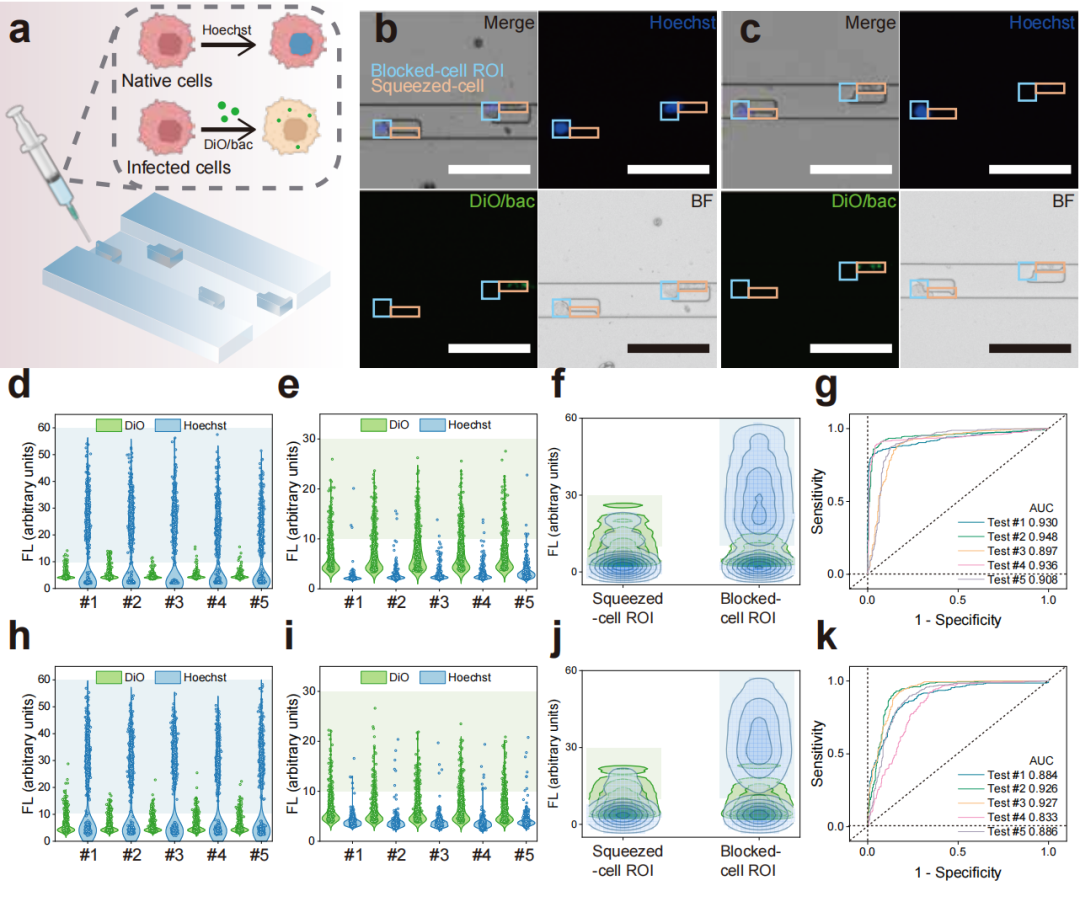
Figure 4: Experimental results of screening bacteria-infected MDA-MB-231 cells using the LesM chip.
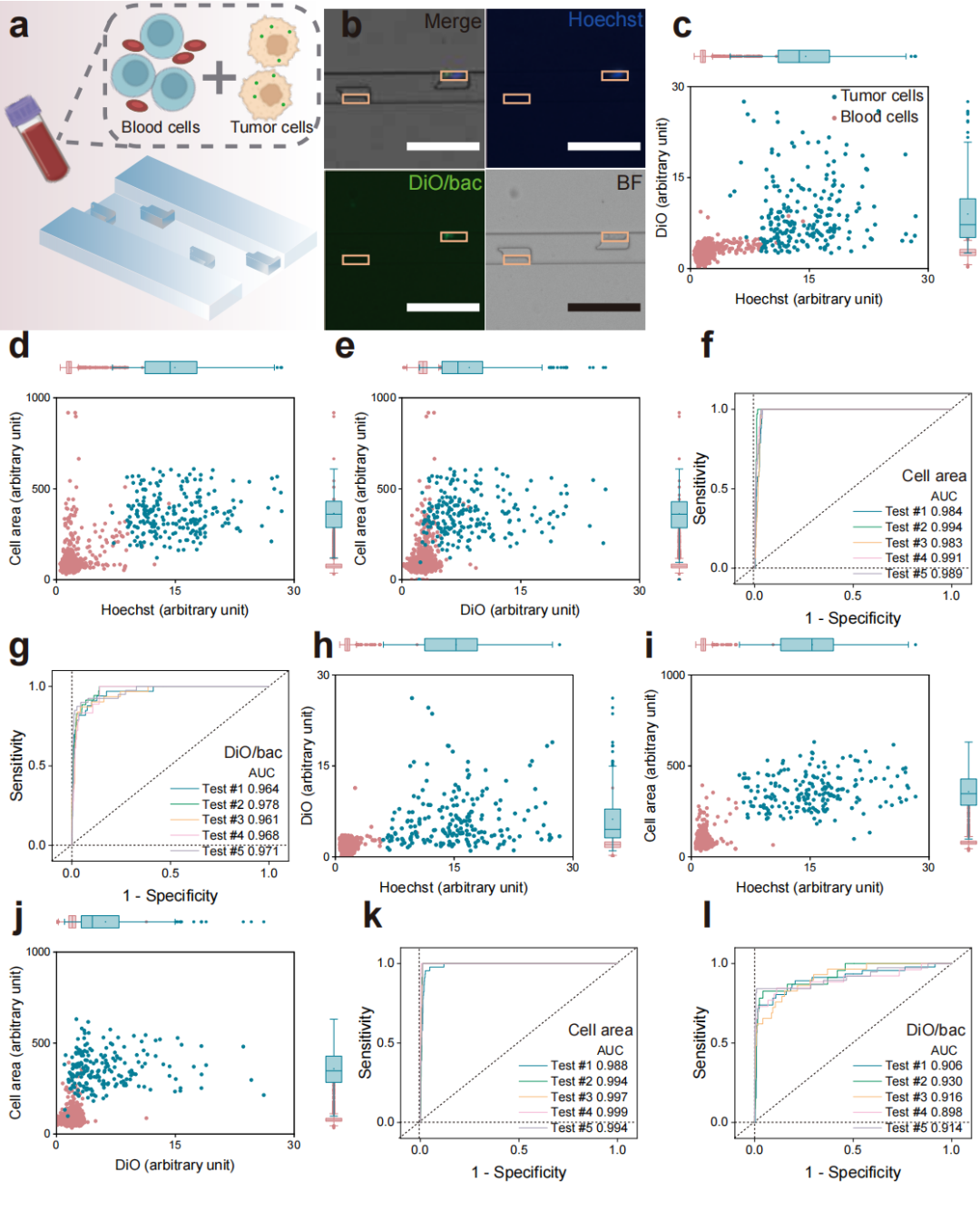
Figure 5: Capacity of the LesM chip to detect bacteria-infected CTCs in whole blood samples.
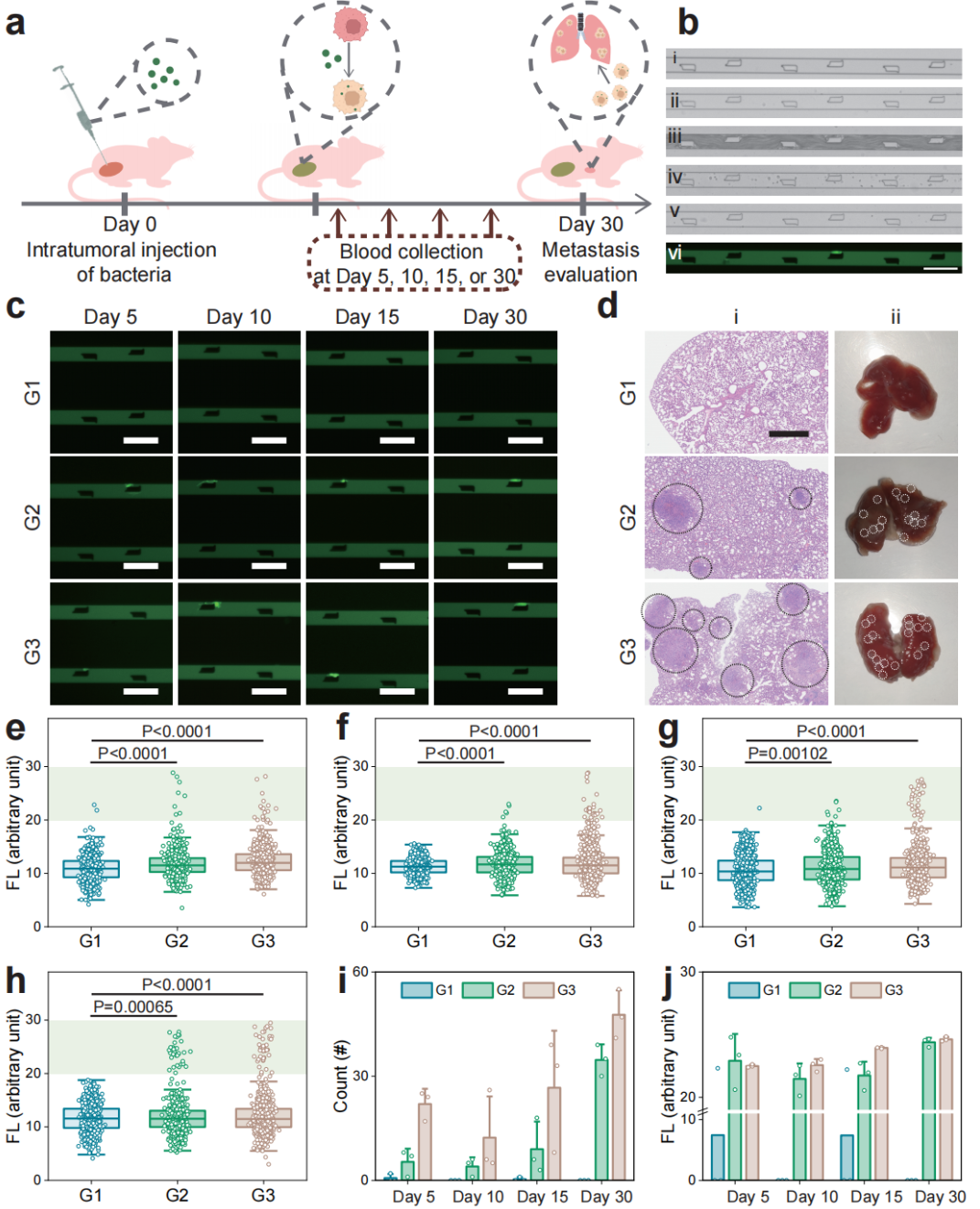
Figure 6: In vivo evaluation of the metastatic potential of bacteria-infected CTCs.
Clinical Value: Initiating a New Paradigm for Precision Cancer Diagnosis and Treatment
The advent of the LesM chip brings multiple breakthroughs for breast cancer metastasis diagnosis and treatment. By simultaneously detecting the mechanical properties and bacterial infection status of circulating tumor cells, it accurately identifies high-metastatic-risk cell subpopulations, addressing the limitation of traditional CTC detection which emphasizes "quantity over quality." It enables direct mechanical screening of unlabeled cells without complex preprocessing, combined with specific fluorescent probe validation, balancing efficiency and accuracy. Furthermore, it allows for evaluating the efficacy of antibiotic therapy, providing a basis for pre-surgical antibacterial intervention to reduce metastasis risk. The study confirmed that pre-surgical antibiotic use significantly reduces the number of infected circulating tumor cells, while post-surgical use is less effective.
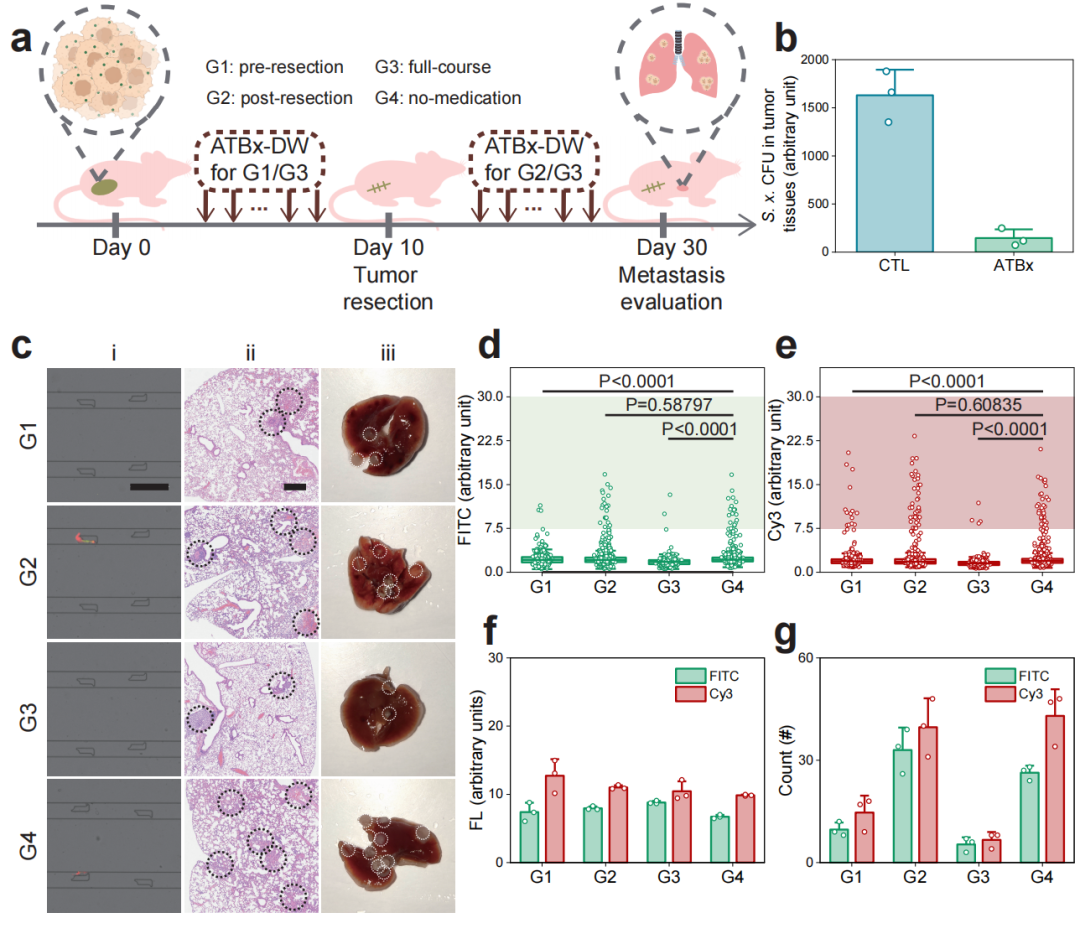
Figure 7: Impact of antibiotic treatment on tumor metastasis.
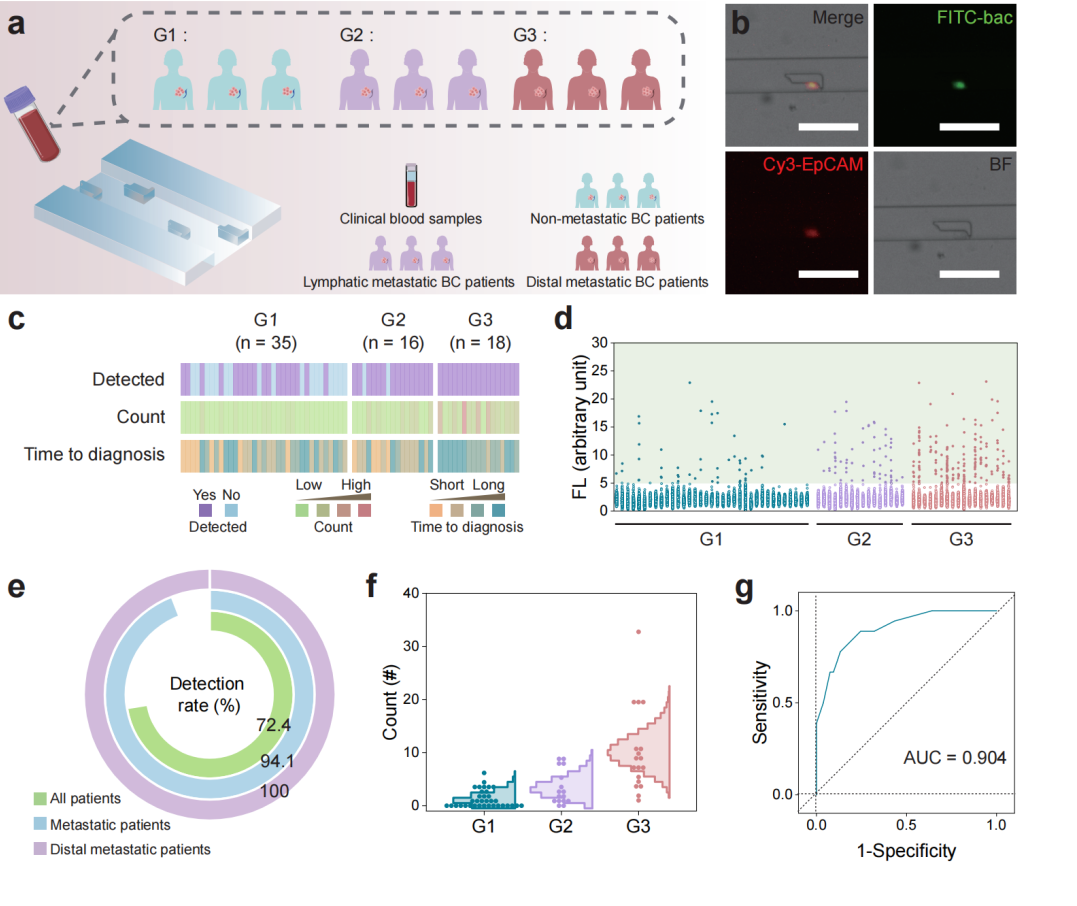
Figure 8: Application of the LesM chip in clinical blood samples from breast cancer patients.
Summary and Outlook
This study successfully developed a microfluidic-based cellular mechanical phenotype screening chip (LesM) capable of efficiently capturing CTCs and distinguishing bacteria-infected cells, providing a new tool for predicting distant metastasis in breast cancer and guiding antibacterial therapy. By combining cell deformability assessment with fluorescence labeling technology, the chip achieves high-sensitivity detection of infected CTCs, and its clinical application value has been validated through in vivo experiments. In the future, this technology holds the potential to be extended to other cancer types, offering stronger support for early cancer diagnosis and personalized treatment. Further research can also explore the impact of a wider range of tumor-associated microorganisms on CTC characteristics to enhance the detection scope and accuracy of this technology.


